Molten Salt vs. Solid-State Synthesis of Gd₂TiO₅: A Comparative Analysis of Efficiency, Phase Purity, and Application Potential
This article provides a comprehensive comparison between molten salt synthesis (MSS) and conventional solid-state synthesis (SSS) for fabricating gadolinium titanate (Gd₂TiO₅), a material of significant interest for nuclear applications and...
Molten Salt vs. Solid-State Synthesis of Gd₂TiO₅: A Comparative Analysis of Efficiency, Phase Purity, and Application Potential
Abstract
This article provides a comprehensive comparison between molten salt synthesis (MSS) and conventional solid-state synthesis (SSS) for fabricating gadolinium titanate (Gd₂TiO₅), a material of significant interest for nuclear applications and advanced ceramics. We explore the foundational principles of Gd₂TiO₅, including its crystal structure and key properties. A detailed methodological analysis contrasts the mechanisms, parameters, and procedural steps of MSS and SSS. The review further addresses common synthesis challenges and optimization strategies, supported by data on phase purity, reaction kinetics, and microstructural characteristics. Aimed at researchers and scientists in materials development, this analysis validates the superior efficiency of MSS and discusses its implications for producing high-performance materials for biomedical and clinical research, such as in radiation shielding or targeted drug delivery systems.
Understanding Gd₂TiO₅: Structure, Properties, and Industrial Significance
Crystal Structure and Polymorphs of Gd₂TiO₅
Gd₂TiO₅ is a complex rare-earth titanate compound of significant interest in materials science, particularly for nuclear applications where it serves as a potential matrix for actinide disposition and as a neutron absorber in control rods [1]. A comprehensive understanding of its crystal structure and the existence of different polymorphic forms is essential for tailoring its properties for specific technological applications. This guide provides a detailed comparison of the identified Gd₂TiO₅ polymorphs, with a specific focus on synthesizing the orthorhombic phase via the increasingly prominent molten salt solid-state synthesis route. The objective analysis presented herein is framed within a broader thesis comparing the efficacy of this method against conventional solid-state synthesis, providing researchers with critical experimental data and protocols.
Crystal Structure and Polymorphs of Gd₂TiO₅
Gd₂TiO₅ exhibits polymorphism, meaning it can crystallize in different structural forms depending on the synthesis conditions and potential elemental substitutions. The most commonly studied and accessed polymorph at room temperature is the orthorhombic structure.
Orthorhombic Gd₂TiO₅ (Primary Polymorph)
The predominant and most stable form of Gd₂TiO₅ under standard conditions is the orthorhombic crystal structure [2] [3]. Key crystallographic parameters for this phase are consolidated in the table below.
Table 1: Crystallographic Parameters for Orthorhombic Gd₂TiO₅
| Parameter | Value | Details |
|---|---|---|
| Pearson Symbol | oP32 | [2] [3] |
| Space Group | 62 (Pnam) | [2] [4] [3] |
| Phase Prototype | La₂TiO₅ | [2] [3] |
| Lattice Parameters | a = 10.460(5) Å, b = 11.317(6) Å, c = 3.750(3) Å | [4] |
| Z (Formula Units/Cell) | 4 | [2] [3] |
| Density | 6.62 Mg·m⁻³ | [2] [3] |
| Determination Method | X-ray diffraction (automatic diffractometer) | [2] |
This structure is characterized by its orthorhombic symmetry and is often the target phase for material synthesis. The Ti and O atoms form a framework where Gd atoms occupy specific interstitial sites, sometimes described as a 'stuffed' pyrochlore derivative [1].
Other Polymorphs
Research indicates that Gd₂TiO₅ can form other metastable or compositionally-induced polymorphs, highlighting the structural diversity within this chemical system.
Table 2: Other Reported Polymorphs in the Ln₂TiO₅ (Ln=Gd, Dy) System
| Polymorph | Composition | Crystal System | Lattice Parameters | Space Group |
|---|---|---|---|---|
| Hexagonal | Gd₁.₈Lu₀.₂TiO₅ | Hexagonal | a = 3.663(3) Å, c = 11.98(1) Å | P6₃/mmc [4] |
| Cubic | Dy₂TiO₅ | Cubic | a = 10.28(1) Å | Fd-3m [4] |
| Monoclinic | Dy₂TiO₅ | Monoclinic | a = 10.33(1) Å, b = 3.653(5) Å, c = 7.306(6) Å, β = 90.00(7)° | B2/m [4] |
The hexagonal phase is stabilized by partial substitution of Gd with Lu, while the cubic and monoclinic phases were identified in dysprosium titanate (Dy₂TiO₅), suggesting that similar polymorphs might be accessible in Gd₂TiO₅ under specific, potentially non-equilibrium, synthesis conditions [4]. The monoclinic polymorph was reported as a new discovery in the cited work.
Synthesis Methodologies: Molten Salt vs. Conventional Solid-State
The synthesis pathway is critical in obtaining phase-pure Gd₂TiO₅ and controlling its polymorphic form. The following section compares the experimental protocols for molten salt and conventional solid-state synthesis.
Experimental Protocols
1. Molten Salt Synthesis (MSS)
This method utilizes a molten salt medium to enhance ion diffusion, thereby accelerating reaction kinetics and lowering the required synthesis temperature and time [1] [5].
- Objective: To synthesize single-phase orthorhombic Gd₂TiO₅ via a low-temperature, rapid route [1] [5].
- Precursors: Gd₂O₃ and TiO₂ (rutile) powder [1]. A variant, the "Firing Salts Method" (FSM), also uses these precursors [5].
- Salt Medium: The specific salt is not detailed in the provided results, but the FSM uses a salt mixture to create the reactive medium [5].
- Procedure:
- The precursor oxides are mixed with an appropriate salt (e.g., NaCl-KCl).
- The mixture is heated to a temperature above the melting point of the salt (e.g., 1200-1300°C) for a short duration (e.g., 2 hours) [1] [5].
- The resulting product is cooled and washed thoroughly with deionized water or a suitable solvent to remove the residual salt, leaving behind the synthesized powder [1].
- Mechanism: The synthesis proceeds through a template growth mechanism, often forming via the initial kinetic product Gd₂Ti₂O₇ (pyrochlore), which subsequently transforms into the 'stuffed' pyrochlore Gd₂TiO₅ [1].
2. Conventional Solid-State Synthesis
This is a standard ceramic method involving direct high-temperature reaction of solid precursors.
- Objective: To synthesize Gd₂TiO₅ through direct solid-state diffusion [6].
- Precursors: Gd₂O₃ and TiO₂ [6].
- Procedure:
- The precursor oxides are thoroughly mixed and ground to ensure homogeneity.
- The mixture is calcined at high temperatures (typically >1300°C) for extended periods (often 12-24 hours) to facilitate the solid-state reaction.
- Intermediate grinding and repeated calcination steps are often required to achieve phase purity.
- Mechanism: Relies on direct solid-state diffusion of cations, which is a slower process compared to solution-based or molten salt methods.
Synthesis Comparison and Workflow
The diagram below illustrates the key steps and comparative advantages of the Molten Salt Synthesis pathway for Gd₂TiO₅.
Figure 1: A comparative workflow of Gd₂TiO₅ synthesis pathways, highlighting the transformative steps in the molten salt route.
Table 3: Quantitative Comparison of Synthesis Methods for Gd₂TiO₅
| Parameter | Molten Salt Synthesis (MSS) | Conventional Solid-State |
|---|---|---|
| Synthesis Temperature | 1200°C [5] - 1300°C [1] | Typically >1300°C [1] |
| Reaction Time | 2 hours [1] [5] | 12-24 hours (often with multiple steps) |
| Key Intermediate | Gd₂Ti₂O₇ Pyrochlore [1] | Not Specified |
| Final Product | Single-phase orthorhombic Gd₂TiO₅ [1] [5] | Orthorhombic Gd₂TiO₅ [6] |
| Particle Size | 50-500 nm (FSM method) [5] | Typically larger, micron-sized |
| Primary Advantage | Lower temperature, faster kinetics, smaller particles | Simple setup, no washing step required |
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful synthesis and characterization of Gd₂TiO₅ require a set of specific reagents and instruments.
Table 4: Essential Materials and Equipment for Gd₂TiO₅ Research
| Item | Function/Description | Reference |
|---|---|---|
| Gadolinium Oxide (Gd₂O₃) | High-purity source of gadolinium cations. | [2] [6] [3] |
| Titanium Dioxide (TiO₂), Rutile | High-purity source of titanium cations. | [2] [3] |
| Molten Salt Medium | Acts as a solvent for oxides, facilitating rapid diffusion and reaction at lower temperatures. | [1] [5] |
| Automatic Diffractometer | Used for determining crystal structure and cell parameters via X-ray diffraction (XRD). | [2] |
| Solid-State Synthesis Furnace | High-temperature furnace capable of sustained operation above 1300°C for conventional synthesis. | [6] |
| High-Resolution TEM | For direct imaging of particle morphology, size distribution, and crystal structure. | [5] |
| Raman Spectrometer | Used for vibrational spectroscopy to confirm chemical bonding and phase identity. | [5] |
Characterization and Functional Properties
The synthesized materials must be rigorously characterized to confirm their structure and evaluate their properties for intended applications.
Characterization Workflow
The typical pathway for validating synthesized Gd₂TiO₅ involves a suite of complementary techniques, as outlined below.
Figure 2: A standard multi-technique characterization workflow for Gd₂TiO₅ materials.
Key Property: Optical Bandgap and Doping Effects
The optical properties of Gd₂TiO₅ are crucial for various functional applications and can be tuned via doping.
- Pure Gd₂TiO₅: The electronic structure near the Fermi level is contributed by Ti-3d and Gd-5d states [6].
- Doping Effects: Substituting cations in Gd₂TiO₅ allows for property engineering:
- Analysis Technique: These observations are supported by optical absorption spectroscopy and first-principles calculations using the GGA+U methodology [6].
This comparison guide delineates the structural and synthetic landscape of Gd₂TiO₅. The orthorhombic structure (Space Group Pnam) is the most prevalent and technologically relevant polymorph. When comparing synthesis strategies, molten salt synthesis demonstrates a clear advantage over conventional solid-state methods by enabling faster reaction times (as little as 2 hours) and lower processing temperatures (1200-1300°C), resulting in phase-pure material with nanoscale particle sizes. This efficiency, coupled with the detailed experimental protocols and characterization workflows provided, positions MSS as a superior synthesis route. These findings reinforce the broader thesis that MSS is a highly effective and efficient methodology for producing complex refractory oxides like Gd₂TiO₅, with significant potential for application in nuclear technology and beyond.
Key Material Properties and Functional Characteristics
The synthesis of advanced ceramic materials, particularly rare-earth titanates, is a critical area of research for applications ranging from solid-state lighting to nuclear energy. Among these materials, gadolinium titanate (Gd₂TiO₅) has emerged as a compound of significant interest due to its unique structural properties and functional characteristics. This comparison guide objectively analyzes Gd₂TiO₅ synthesized via molten salt synthesis (MSS) in comparison to conventional solid-state synthesis (SSS) and other related perovskite materials. The evaluation is framed within the broader context of optimizing synthesis protocols for enhanced material performance, providing researchers with critical data for selecting appropriate synthesis methodologies based on their specific application requirements.
Synthesis Methodologies: A Comparative Analysis
Fundamental Synthesis Approaches
The synthesis of Gd₂TiO₅ can be achieved through multiple pathways, each with distinct advantages and limitations:
Molten Salt Synthesis (MSS) utilizes a eutectic mixture of salts (typically NaCl:KCl) that melts at relatively low temperatures to create a liquid medium that enhances reactant diffusion. This method operates through a "template growth" mechanism where one reactant dissolves into the salt medium and diffuses onto the surface of the less soluble reactant to form the target product [7]. The process typically employs a 7:1 molar ratio of molten salt to ceramic precursor and can achieve phase purity at significantly lower temperatures and shorter durations than conventional methods.
Conventional Solid-State Synthesis (SSS) relies on direct reaction between metal oxide precursors (Gd₂O₃ and TiO₂) through repeated milling and high-temperature treatments to facilitate solid-state diffusion. This method typically requires multiple processing steps with extended dwell times at elevated temperatures to achieve complete reaction [7].
Firing Salts Method (FSM) represents a variation on MSS, optimized to produce orthorhombic Gd₂TiO₅ at 1200°C with just a 2-hour dwell time. This method results in particle size distributions between 50-500 nm and has been characterized extensively through X-ray diffraction, high-resolution transmission electron microscopy, and vibrational spectroscopy [8].
Comparative Performance Metrics
Table 1: Quantitative Comparison of Gd₂TiO₅ Synthesis Methods
| Synthesis Parameter | Molten Salt Synthesis | Conventional Solid-State Synthesis | Firing Salts Method |
|---|---|---|---|
| Optimal Temperature | 1300°C | >1500°C | 1200°C |
| Reaction Time | 2 hours | 24+ hours (multiple steps) | 2 hours |
| Phase Purity | Single phase achieved | Trace impurities persist | Orthorhombic phase achieved |
| Particle Size | ~1 μm (facetted morphology) | 2-3 μm (angular morphology) | 50-500 nm |
| Mechanism | Template growth | Solid-state diffusion | Modified salt-assisted |
| Energy Consumption | Lower | Higher | Moderate |
Table 2: Comparison of Gd₂TiO₅ with Related Materials for Solid-State Lighting
| Material Property | Gd₂TiO₅:Eu³⁺ | GdAlO₃:Cr³⁺/Sm³⁺ | Conventional Phosphors |
|---|---|---|---|
| Crystal Structure | Orthorhombic | Orthorhombic perovskite | Varies |
| Dopant Solubility | Up to 10 mol% Eu³⁺ without quenching | ~0.5% for both Cr³⁺ and Sm³⁺ | Typically <5% |
| Photoluminescence Quantum Yield | ~14% | Not specified | Varies widely |
| Color Purity | ~99% (red emission) | Energy transfer between ions | Dependent on composition |
| Primary Applications | pc-wLEDs, nuclear | Solid-state lighting | Lighting, displays |
Experimental Protocols and Characterization
Synthesis Workflow
The following diagram illustrates the comparative synthesis pathways for Gd₂TiO₅ production:
Comparative Synthesis Workflow for Gd₂TiO₅ Production
Detailed Experimental Protocols
Molten Salt Synthesis Protocol
The MSS of Gd₂TiO₅ begins with drying Gd₂O₃ at 800°C and TiO₂ at 180°C overnight to remove adsorbed moisture [7]. Stoichiometric amounts of these precursors are batched with a eutectic NaCl:KCl salt mixture (1:1 molar ratio) at a 7:1 molar ratio of salt to ceramic precursor. The mixture is minimally milled at 250 rpm for 5 minutes using cyclohexane as a carrier fluid to prevent excessive size reduction. The resulting mixture is uniaxially pressed into pellets at 3 tonnes for 1 minute, then reacted at 1300°C for 2 hours with heating and cooling rates of 5°C/min. The product is washed with ultrapure water to remove the salt medium and collected via vacuum filtration [7].
Solid-State Synthesis Protocol
For SSS, stoichiometric Gd₂O₃ and TiO₂ are planetary ball milled at 500 rpm for 15 minutes using isopropanol as the carrier fluid [7]. The slurry is dried at 80°C and sieved through a 250 μm mesh. The powder is pressed into pellets under the same conditions as MSS, but reacted at temperatures ranging from 700-1500°C for 24 hours at each temperature step. Multiple heating steps with intermediate milling are typically required to achieve homogeneity, and trace impurities often persist even after high-temperature treatment [7].
Dopant Incorporation for Optical Applications
For photoluminescent applications such as Eu³⁺ doping, the synthesis follows similar protocols with the addition of Eu₂O₃ to the precursor mixture [9]. The nominal composition Gd₂₋ₓEuₓTiO₅ (where x = 0.00-0.20) is processed with three-step heating protocols to ensure complete reaction. The doped materials undergo additional characterization including photoluminescence spectroscopy, Judd-Ofelt analysis, and positron annihilation lifetime spectroscopy to evaluate optical performance and defect structures [9].
Characterization Techniques
Comprehensive characterization is essential for evaluating material properties:
Structural Analysis: Powder X-ray diffraction (XRD) with Rietveld refinement confirms phase purity and crystal structure. Gd₂TiO₄ crystallizes in an orthorhombic structure (Pnma) comprising edge-sharing GdO₇ polyhedra with Ti cations forming TiO₅ square pyramids [7].
Morphological Evaluation: Scanning electron microscopy (SEM) reveals particle morphology and size distribution, showing MSS produces faceted particles of ~1 μm while SSS yields larger, angular particles of 2-3 μm [7].
Optical Characterization: For doped materials, photoluminescence excitation and emission spectra, decay lifetime measurements, and determination of absolute photoluminescence quantum yield (PLQY) are essential. Diffuse reflectance spectroscopy provides band gap information [9].
Defect Analysis: Positron annihilation lifetime spectroscopy (PALS) probes vacancy-type defects and their evolution with doping concentration, providing critical insights into charge compensation mechanisms [9].
Research Reagent Solutions
Table 3: Essential Research Reagents for Gd₂TiO₅ Synthesis
| Reagent/Material | Specifications | Function in Synthesis |
|---|---|---|
| Gadolinium Oxide (Gd₂O₃) | 99.9% purity, pre-dried at 800°C | Primary rare-earth cation source for host lattice |
| Titanium Dioxide (TiO₂) | 99.0% purity, pre-dried at 180°C | Transition metal oxide source for structural framework |
| Europium Oxide (Eu₂O₃) | 99.0% purity, pre-dried at 800°C | Luminescent dopant for red emission in phosphor applications |
| Sodium Chloride (NaCl) | High purity, anhydrous | Component of eutectic salt mixture for MSS |
| Potassium Chloride (KCl) | High purity, anhydrous | Component of eutectic salt mixture for MSS |
| Isopropanol | Laboratory grade | Carrier fluid for milling in SSS |
| Cyclohexane | Laboratory grade | Carrier fluid for milling in MSS |
Functional Performance in Applications
Optical Performance Metrics
Gd₂TiO₅:Eu³⁺ demonstrates exceptional properties for solid-state lighting applications, exhibiting no concentration quenching up to 10 mol% doping - a significant advantage over many alternative phosphor materials [9]. The material maintains high color purity (~99%) and photoluminescence quantum yield (~14%) while showing monotonic rise in emission intensity with increasing dopant concentration [9]. The stability of Eu³⁺ local symmetry and Eu-O bond polarizability throughout the concentration range indicates minimal structural distortion with doping.
The comparative energy transfer mechanisms in related materials further highlight the unique advantages of Gd₂TiO₅ as a host material. In Cr³⁺/Sm³⁺ doped GdAlO₃ perovskite, energy transfer occurs between Gd³⁺, Sm³⁺ and Cr³⁺ ions, but with significantly lower dopant solubility (~0.5% for each ion) compared to the high solubility achievable in Gd₂TiO₅ [10].
Structural and Defect Characteristics
The unusual coordination polyhedra in Gd₂TiO₅ - including five-coordinated Ti⁴⁺ (TiO₅) and seven-coordinated Gd³⁺ (GdO₇) - create a rigid structural framework that enables high dopant incorporation without significant lattice strain [9]. Positron annihilation lifetime spectroscopy confirms that average positron lifetime remains constant across doping concentrations, indicating no charge-compensating defect formation and justifying isovalent occupancy of Eu³⁺ at Gd³⁺ sites [9].
This defect tolerance represents a significant advantage over many alternative host materials where charge compensation mechanisms often lead to vacancy formation that promotes concentration quenching and reduces luminescence efficiency.
This comparison guide demonstrates that Gd₂TiO₅ synthesized via molten salt methods offers distinct advantages over conventional solid-state synthesis and related materials for advanced applications. The MSS approach enables lower processing temperatures, shorter reaction times, and improved phase purity compared to SSS, while the unique structural characteristics of Gd₂TiO₅ support high dopant solubility and exceptional optical properties. When doped with Eu³⁺, this material system achieves remarkable performance metrics including minimal concentration quenching, high color purity, and substantial quantum yield. These characteristics position Gd₂TiO₅ as a highly promising material for phosphor-converted white light-emitting diodes and nuclear applications, with MSS representing the most efficient synthesis pathway for producing high-quality material with controlled morphology and properties.
Primary Applications in Nuclear and Advanced Technologies
Gadolinium titanate (Gd₂TiO₅) represents a significant material in the realm of advanced ceramics, finding critical applications across nuclear energy and solid-state lighting technologies. This refractory material belongs to the class of 'stuffed' pyrochlores, characterized by its orthorhombic crystal structure (space group Pnma) comprising a framework of edge-sharing GdO₇ polyhedra with Ti cations occupying interstices within the tunnels, forming TiO₅ square pyramids [11] [7]. The material's value stems from its exceptional thermal stability, chemical resistance, and structural flexibility, allowing for extensive doping with various lanthanide ions [11] [9].
The synthesis pathway for Gd₂TiO₅ significantly influences its material properties and ultimate performance in applications. While conventional solid-state synthesis (SSS) has been widely used, molten salt synthesis (MSS) has emerged as a superior alternative for specific technological needs. SSS typically involves high-temperature reactions (≥1300°C) for extended durations (24 hours or more) through solid-state diffusion of oxide precursors (Gd₂O₃ and TiO₂) [7] [9]. In contrast, MSS utilizes a molten salt medium (e.g., NaCl:KCl eutectic) at lower temperatures (650-1300°C) for significantly shorter durations (1-2 hours), enabling rapid diffusion of reactants and formation of phase-pure products with controlled morphology [7] [12]. This comprehensive analysis compares these synthesis methodologies, providing experimental data and protocols to guide researchers in selecting optimal synthesis routes for nuclear and advanced technology applications.
Comparative Analysis of Synthesis Methods
Performance and Outcome Comparison
Table 1: Direct comparison between Molten Salt Synthesis and Solid-State Synthesis for Gd₂TiO₅
| Parameter | Molten Salt Synthesis (MSS) | Conventional Solid-State Synthesis (SSS) |
|---|---|---|
| Synthesis Temperature | 650-1300°C [7] [12] | 1300-1500°C [7] [9] |
| Reaction Time | 1-2 hours [7] [12] | 24+ hours [7] [9] |
| Phase Purity | Single-phase achievable at 1300°C/2h [7] | Trace impurities (Gd₂Ti₂O₇, Gd₂O₃) even at 1500°C [7] |
| Particle Morphology | Template growth mechanism, similar to TiO₂ precursor (~1 μm) [7] | Larger particles (2-4 μm), angular morphology [7] |
| Particle Size Control | Excellent via precursor selection [7] | Limited, larger aggregates [7] |
| Energy Consumption | Lower (reduced T/t) [7] | Higher (elevated T/t) [7] |
| Scalability | High, suitable for powder production [7] [12] | Moderate, requires repeated milling/calcination [7] |
| Dopant Incorporation | Effective for lanthanides [12] | Effective, but may require higher temperatures [9] |
Application-Specific Performance Metrics
Table 2: Application-driven performance metrics of Gd₂TiO₅ synthesized via different methods
| Application | Synthesis Method | Key Performance Metrics | Experimental Results |
|---|---|---|---|
| Nuclear Waste Forms [7] [12] | MSS (CaCl₂:NaCl) | Phase purity, actinide incorporation | Single-phase Gd₂TiO₅ at 650°C/2h; Effective Sm³+ doping as actinide surrogate [12] |
| Solid-State Lighting [11] [9] | Sol-gel (modified MSS) | Quantum efficiency, color purity | Gd₂TiO₅:Eu³+ - PLQY 61%; Color purity ~99% [11] [9] |
| Solid-State Lighting [11] | Sol-gel | White light emission, photostability | Gd₂TiO₅:Dy³+/Eu³+ - Tunable white emission; Outstanding photostability [11] |
| Neutron Absorbers [7] | SSS | Phase stability, structural integrity | Gd₂TiO₅ formation at 1500°C with trace impurities [7] |
Experimental Protocols and Methodologies
Molten Salt Synthesis Protocol
Reagents and Equipment:
- Gd₂O₃ (pre-dried at 800°C overnight)
- TiO₂ (pre-dried at 180°C overnight)
- NaCl:KCl eutectic mixture (1:1 molar ratio)
- High-temperature furnace (>1300°C capability)
- Planetary ball mill (Pulverisette 6, Fritsch or equivalent)
- Vacuum filtration apparatus
Step-by-Step Procedure:
- Precursor Preparation: Dry Gd₂O₃ at 800°C and TiO₂ at 180°C overnight to remove adsorbed moisture [7].
- Stoichiometric Batching: Weigh stoichiometric amounts of Gd₂O₃ and TiO₂ for 10 g batch of Gd₂TiO₅ [7].
- Salt Addition: Add NaCl:KCl eutectic mixture at 7:1 molar ratio of molten salt to ceramic precursors [7].
- Milling: Mill the salt and precursor mixture at 250 rpm for 5 minutes using cyclohexane as carrier fluid to minimize size reduction [7].
- Pellet Formation: Uniaxially press 0.5 g samples as 13 mm pellets at 3 tonnes for 1 minute [7].
- Thermal Treatment: Heat pellets at 5°C/min to 1300°C, hold for 2 hours, then cool at 5°C/min [7].
- Salt Removal: Wash the reacted product with ultrapure water to remove molten salt medium, collect powder via vacuum filtration [7].
- Characterization: Validate phase purity by X-ray diffraction, comparing to ICDD PDF-4+ 21-0342 [7].
Critical Parameters for Success:
- The 7:1 salt-to-precursor ratio is optimal for complete reaction [7]
- Heating/cooling rates of 5°C/min prevent thermal shock and maintain structural integrity [7]
- Complete salt removal is verified by absence of NaCl/KCl XRD reflections [7]
Conventional Solid-State Synthesis Protocol
Reagents and Equipment:
- Gd₂O₃ (pre-dried at 800°C overnight)
- TiO₂ (pre-dried at 180°C overnight)
- Isopropanol (carrier fluid)
- High-temperature furnace (>1500°C capability)
- Planetary ball mill with Sialon pot and media
- 250 μm sieve
Step-by-Step Procedure:
- Precursor Preparation: Dry Gd₂O₃ at 800°C and TiO₂ at 180°C overnight [7] [9].
- Stoichiometric Weighing: Batch stoichiometric amounts of reagents for 10 g of Gd₂TiO₅ [7].
- Intensive Milling: Planetary ball mill at 500 rpm for 15 minutes using isopropanol as carrier fluid [7].
- Drying and Sieving: Dry resulting powder slurry at 80°C overnight and pass through 250 μm sieve [7].
- Pellet Formation: Uniaxially press 0.5 g samples as 13 mm pellets at 3 tonnes for 1 minute [7].
- Multi-Step Calcination: Heat pellets at 5°C/min to 700-1500°C in 100°C intervals, holding for 24 hours at each temperature [7]. Alternatively, single-step reaction at 1300°C for 24 hours [9].
- Characterization: Validate phase composition by X-ray diffraction; note that trace Gd₂Ti₂O₇ and Gd₂O₃ may persist even at 1500°C [7].
Optimization Notes:
- Multiple heating steps with intermediate grinding may improve phase purity [7]
- For doped compositions (e.g., Gd₂₋ₓEuₓTiO₅), three-step heating protocol is recommended [9]
Synthesis Mechanisms and Pathways
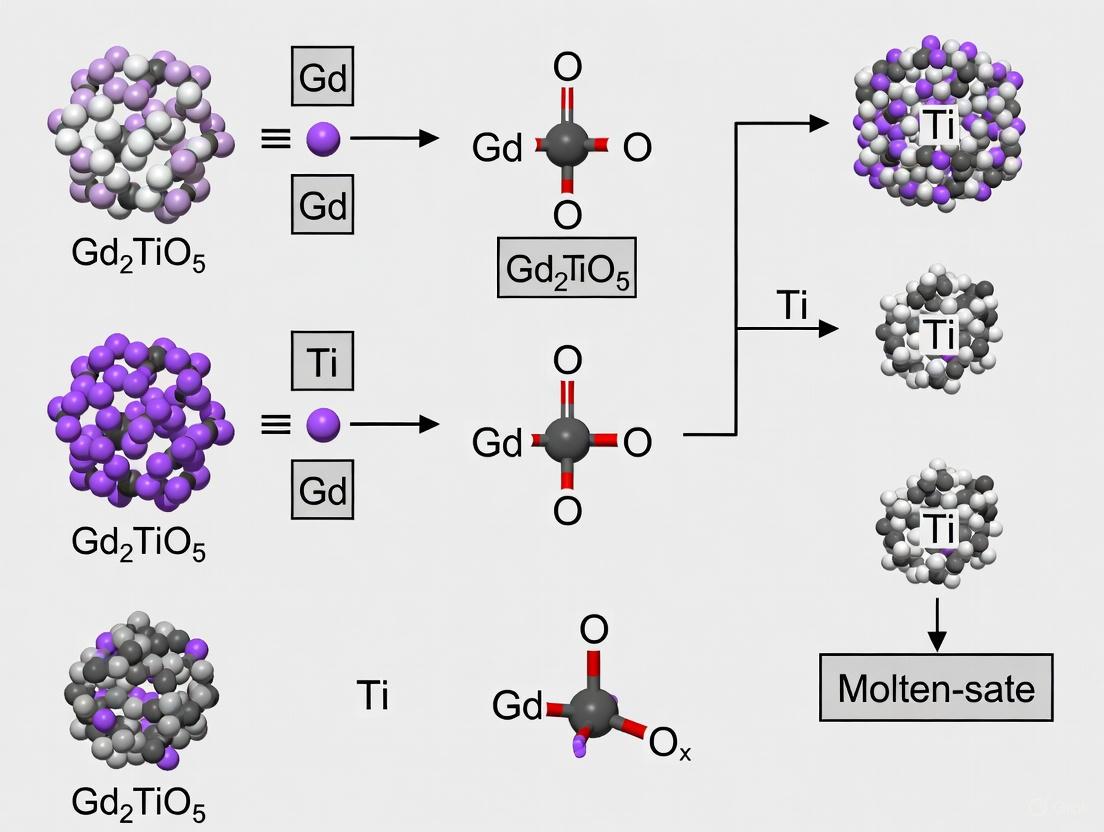
The synthesis mechanisms differ fundamentally between MSS and SSS approaches. MSS proceeds primarily through a template growth mechanism (solution-diffusion), where TiO₂ particles act as templates with lower dissolution rates, while Gd₂O₃ dissolves more readily into the molten salt medium [7] [12]. The dissolved Gd species diffuse through the salt to react with TiO₂ templates, forming Gd₂TiO₅ while preserving the original TiO₂ morphology [7]. This explains the similar particulate morphology and dimensions (~1 μm) observed between the TiO₂ reagent and MSS product [7].
In contrast, SSS relies exclusively on solid-state diffusion, where intimate mixing of precursors enhances contact points, but atomic diffusion occurs only at elevated temperatures (>1300°C) over extended time periods [7] [9]. This process involves repeated milling and calcination cycles to improve homogeneity, but often results in larger particles (2-4 μm) with incomplete reaction, evidenced by persistent minor phases (Gd₂Ti₂O₇, Gd₂O₃) even after 24 hours at 1500°C [7].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential research reagents and materials for Gd₂TiO₅ synthesis
| Reagent/Material | Specifications | Function in Synthesis | Application Notes |
|---|---|---|---|
| Gadolinium Oxide (Gd₂O₃) | 99.9% purity, pre-dried at 800°C [7] [9] | Gd source for Gd₂TiO₅ matrix | Moisture-sensitive; requires pre-drying to remove adsorbed H₂O/CO₂ [7] |
| Titanium Dioxide (TiO₂) | 99.0-99.995% purity, pre-dried at 180°C [7] [9] | Ti source for Gd₂TiO₅ matrix | Anatase or rutile phase; particle size determines product morphology in MSS [7] |
| Sodium Chloride (NaCl) | 99.9% purity, dried at 110°C [7] | Component of eutectic salt medium | Forms low-melting eutectic with KCl (mp ~650°C) [7] [12] |
| Potassium Chloride (KCl) | 99.9% purity, dried at 110°C [7] | Component of eutectic salt medium | Forms low-melting eutectic with NaCl; creates liquid reaction medium [7] [12] |
| Calcium Chloride (CaCl₂) | Anhydrous, 99% purity [12] | Alternative salt medium for nuclear applications | Compatible with Am extraction process salts; lower melting eutectics [12] |
| Europium Oxide (Eu₂O₃) | 99.9% purity, pre-dried at 800°C [11] [9] | Dopant for photoluminescence | Creates red emission centers; complete solubility in Gd₂TiO₅ up to 10 mol% [9] |
| Dysprosium Nitrate (Dy(NO₃)₃·xH₂O) | 99.9% purity [11] | Dopant for white light emission | Combined with Eu³+ creates tunable white emission for pc-WLEDs [11] |
The comparative analysis demonstrates that MSS offers significant advantages over SSS for Gd₂TiO₅ synthesis, particularly in nuclear applications where low-temperature processing of actinide-containing wastes is essential [7] [12]. MSS achieves single-phase Gd₂TiO₅ at 1300°C in 2 hours, while SSS requires higher temperatures (1500°C) and longer durations (24+ hours) while still retaining trace impurities [7]. The template growth mechanism in MSS enables superior morphological control, producing finer particles (~1 μm) compared to SSS (2-4 μm) [7].
For photoluminescence applications, both synthesis methods effectively incorporate lanthanide dopants (Eu³+, Dy³+), enabling the development of efficient phosphors for solid-state lighting [11] [9]. Gd₂TiO₅:Eu³+ exhibits exceptional color purity (~99%) and photoluminescence quantum yield (61%) [11] [9], while Dy³+/Eu³+ co-doping enables tunable white emission with outstanding photostability [11]. The choice between MSS and SSS for lighting applications depends on specific morphological requirements and processing constraints.
Future research directions should explore MSS with alternative salt eutectics for lower synthesis temperatures, investigate radiation tolerance of MSS-derived Gd₂TiO₅ for nuclear applications, and optimize dopant combinations for specialized lighting needs. The demonstrated efficacy of MSS positions it as the preferred synthesis method for next-generation Gd₂TiO₅ materials across nuclear and advanced technology applications.
The Critical Need for Efficient Synthesis Pathways
The pursuit of advanced ceramic materials for demanding applications, such as nuclear reactors and solid-state lighting, has intensified the focus on efficient and reliable synthesis pathways. Among these materials, gadolinium titanate (Gd₂TiO₅), a refractory 'stuffed' pyrochlore, has garnered significant interest due to its potential use as a matrix for actinide disposition and as a neutron absorber in control rods [7]. Its utility extends to the field of photonics, where it serves as a host lattice for single-component white-emitting nanophosphors [11]. The synthesis of such a refractory compound, however, presents considerable challenges. Conventional solid-state reactions often involve high temperatures and prolonged durations, leading to coarse grains and potential non-stoichiometry due to volatile component loss [7]. This article provides a comparative guide examining the performance of conventional solid-state synthesis against the Molten Salt Synthesis (MSS) route for Gd₂TiO₅, presenting objective experimental data to underscore the critical advantages of MSS in modern materials research.
Synthesis Methodologies: A Detailed Comparison
The synthesis of Gd₂TiO₅ can be achieved through multiple pathways, each with distinct protocols and outcomes. The following sections detail the methodologies of two primary techniques: conventional Solid-State Synthesis (SSS) and Molten Salt Synthesis (MSS).
Solid-State Synthesis (SSS) Protocol
The conventional Solid-State Synthesis method is a widely used ceramic processing technique. A typical experimental protocol is as follows [7]:
- Precursor Preparation: Gd₂O₃ and TiO₂ reagents are dried overnight at 800 °C and 180 °C, respectively. Stoichiometric amounts are batched.
- Milling: The powder mixture is planetary ball-milled (e.g., at 500 rpm for 15 minutes) using a carrier fluid like isopropanol to ensure homogenization.
- Pelletization: The resulting slurry is dried and sieved, after which powder is uniaxially pressed into pellets.
- Reaction: The green pellets are subjected to high-temperature reactions, typically in the range of 700–1500 °C for 24 hours at each target temperature, with controlled heating and cooling rates (e.g., 5 °C/min).
Molten Salt Synthesis (MSS) Protocol
The Molten Salt Synthesis method utilizes a salt medium to enhance diffusion. A representative protocol for Gd₂TiO₅ is outlined below [7]:
- Precursor and Salt Preparation: Gd₂O₃ and TiO₂ are dried as in the SSS method. An equimolar eutectic mixture of NaCl and KCl (melting point ~657 °C) is used as the flux, dried at 110 °C.
- Mixing: The salt and precursor oxides are mixed at a 7:1 molar ratio of salt to ceramic precursor.
- Milling: The combined mixture is milled at a lower energy input (e.g., 250 rpm for 5 minutes) to minimize size reduction, using a carrier fluid like cyclohexane.
- Reaction: The pellets are reacted at temperatures between 700–1500 °C for only 2 hours, with the same 5 °C/min ramp rates.
- Purification: The cooled product is washed with ultrapure water to dissolve and remove the salt flux, and the resulting powder is collected via vacuum filtration.
Performance Comparison: Experimental Data
The efficacy of these synthesis methods is objectively compared using key performance metrics, including phase purity, processing conditions, and particle morphology. The quantitative data summarized in the table below highlights the stark differences between the two approaches.
Table 1: Comparative Performance of SSS and MSS for Gd₂TiO₅ Synthesis
| Performance Metric | Solid-State Synthesis (SSS) | Molten Salt Synthesis (MSS) |
|---|---|---|
| Typical Reaction Temperature | 1300-1500 °C | 1200-1300 °C [7] [8] |
| Typical Reaction Time | 24 hours or more [7] | 2 hours [7] |
| Time to Single Phase (at 1300°C) | Not achieved; trace impurities persist even after 24h at 1500°C [7] | Achieved in 2 hours [7] |
| Primary Mechanism | Solid-state diffusion [7] | Template growth (Solution-Diffusion) [7] |
| Resulting Particle Morphology | Larger (2-3 μm), angular particles [7] | Smaller (~1 μm), faceted particles resembling TiO₂ precursor morphology [7] |
| Scalability & Energy Consumption | High energy consumption due to prolonged high-temperature treatment [7] | Lower energy consumption; highly scalable production process [13] |
The data demonstrates that MSS offers a dramatic reduction in reaction time—from 24 hours to just 2 hours—while simultaneously achieving superior phase purity at a lower overall energy cost. The MSS process also enables better control over particle size and morphology, a critical factor for subsequent processing and application performance.
The Scientist's Toolkit: Essential Research Reagents
The successful synthesis of Gd₂TiO₅, particularly via the MSS route, relies on a specific set of reagents and equipment. The following table details these essential materials and their functions.
Table 2: Key Research Reagent Solutions for Gd₂TiO₅ Synthesis
| Reagent / Equipment | Function in the Synthesis Process |
|---|---|
| Gadolinium Oxide (Gd₂O₃) | High-purity (>99%) source of Gd cations; requires pre-drying at 800°C [7]. |
| Titanium Dioxide (TiO₂) | High-purity (>99%) source of Ti cations; requires pre-drying at 180°C [7]. |
| Eutectic NaCl:KCl Salt | Molten salt flux that creates a liquid medium to enhance ionic diffusion and facilitate a lower temperature reaction [7] [14]. |
| Planetary Ball Mill | Equipment used for homogenizing precursor mixtures and for intimately mixing oxide precursors with the salt flux [7]. |
| Vacuum Filtration Setup | Critical for the post-synthesis purification step in MSS, allowing for the efficient separation of the synthesized powder from the water-soluble salt flux [7]. |
Underlying Mechanisms and Workflows
The stark differences in outcomes between SSS and MSS can be traced to their fundamental reaction mechanisms. The following diagram illustrates the comparative workflow and the governing mechanism of the superior MSS pathway.
Diagram 1: Synthesis Workflow Comparison
The MSS method operates primarily via a template growth mechanism (a form of solution-diffusion), which is pivotal to its efficiency. This process is detailed below.
Diagram 2: MSS Template Growth Mechanism
The experimental data and comparative analysis presented in this guide unequivocally demonstrate that Molten Salt Synthesis offers a superior pathway for the synthesis of Gd₂TiO₅ compared to conventional solid-state methods. The key advantages of MSS—significantly shorter reaction times, lower processing temperatures, superior phase purity, and finer, more controlled particle morphology—address the critical need for efficient synthesis pathways in modern materials science. For researchers and industries focused on advanced ceramics for nuclear energy, photonics, and other high-technology fields, the adoption of MSS represents a strategic step towards more sustainable, economical, and high-performance material fabrication.
Synthesis in Action: Step-by-Step Protocols for MSS and SSS
The pursuit of efficient and high-quality synthetic routes for advanced ceramic materials is a central theme in materials science. For refractory compounds such as Gd₂TiO₅ 'stuffed' pyrochlore, the choice of synthesis method profoundly influences critical characteristics including phase purity, particle morphology, reaction kinetics, and ultimately, the material's suitability for advanced applications in nuclear energy and solid-state lighting. This guide provides a detailed comparative analysis of two prominent synthesis techniques: the conventional Solid-State Synthesis (SSS) and the alternative Molten Salt Synthesis (MSS). The SSS method represents the traditional approach, relying on high temperatures and prolonged reaction times to facilitate solid-state diffusion between metal oxide precursors. In contrast, MSS utilizes a molten salt medium to accelerate diffusion and reaction kinetics, often resulting in lower processing temperatures, shorter reaction durations, and superior control over the final product's characteristics. By objectively examining experimental data and underlying mechanisms, this comparison aims to equip researchers with the necessary information to select the optimal synthesis pathway for their specific research and development goals.
Comparative Experimental Synthesis of Gd₂TiO₅
Solid-State Synthesis (SSS) Protocol
The conventional Solid-State Synthesis of Gd₂TiO₅ follows a multi-step procedure that emphasizes mechanical mixing and high-temperature treatment. The protocol, as detailed in a comparative investigation, is as follows [7]:
- Precursor Preparation: Gd₂O₃ and TiO₂ reagents are first dried at 800 °C and 180 °C, respectively, overnight to remove any adsorbed moisture.
- Stoichiometric Batching: Precursors are weighed in stoichiometric proportions to yield a 10-gram batch of Gd₂TiO₅.
- Mechanical Milling: The batched powder is subjected to planetary ball milling at 500 rpm for 15 minutes, using isopropanol as a carrier fluid to ensure homogeneous mixing. The resulting slurry is then dried at 80 °C and passed through a 250 μm sieve.
- Pelletization: The mixed powder is uniaxially pressed into 13 mm pellets under a load of 3 tonnes for 1 minute to form "green" pellets, which enhances interparticle contact during the reaction.
- High-Temperature Reaction: The pellets are reacted in a furnace at temperatures ranging from 700 °C to 1500 °C (in 100 °C intervals) for a duration of 24 hours at each temperature. The heating and cooling rates are maintained at 5 °C per minute.
A significant limitation of this method is that even at 1500 °C, trace amounts of intermediate and precursor phases (Gd₂Ti₂O₇ and Gd₂O₃) can persist, necessitating additional heat treatments to achieve a single-phase product [7].
Molten Salt Synthesis (MSS) Protocol
The Molten Salt Synthesis method introduces a liquid reaction medium to facilitate the formation of Gd₂TiO₅. The established protocol is outlined below [7]:
- Precursor and Salt Preparation: The same Gd₂O₃ and TiO₂ precursors are dried identically to the SSS method. An equimolar eutectic mixture of NaCl and KCl is used as the molten salt flux, which is dried at 110 °C.
- Batching and Low-Energy Milling: Stoichiometric precursor powders are batched for a 10-gram product. The salt and precursor mixture is combined at a 7:1 molar ratio of salt to ceramic product. This mixture is milled at a lower energy (250 rpm for 5 minutes) using cyclohexane as the carrier fluid to minimize excessive size reduction of the reagents.
- Pelletization and Reaction: The mixture is pressed into pellets under identical conditions as the SSS route. The pellets are then reacted at temperatures from 700 °C to 1500 °C for a significantly shorter duration of 2 hours at each temperature, with the same 5 °C/min heating and cooling rates.
- Salt Removal: After the reaction, the cooled product is washed with ultrapure water to dissolve and remove the molten salt flux. The final powder is collected via vacuum filtration.
This method has been demonstrated to yield single-phase Gd₂TiO₅ at 1300 °C after only 2 hours of reaction time [7].
Table 1: Comparative Summary of Synthesis Protocols for Gd₂TiO₅
| Parameter | Solid-State Synthesis (SSS) | Molten Salt Synthesis (MSS) |
|---|---|---|
| Reaction Medium | Solid (no medium) | Liquid (NaCl:KCl eutectic) |
| Milling Intensity | High-energy (500 rpm, 15 min) | Low-energy (250 rpm, 5 min) |
| Typical Reaction Temperature | 1500 °C | 1300 °C |
| Typical Reaction Time | 24 hours | 2 hours |
| Post-Synthesis Processing | None | Washing with water to remove salt |
| Key Mechanism | Solid-state diffusion | Template growth / Solution-precipitation |
Reaction Pathways and Phase Evolution
The formation of Gd₂TiO₅ does not occur via a direct reaction between Gd₂O₃ and TiO₂. Instead, both synthesis methods share a common reaction pathway, albeit with vastly different kinetics [7] [1].
The initial kinetic product is the pyrochlore phase Gd₂Ti₂O₇. This intermediate then reacts with additional Gd₂O₃ to form the final 'stuffed' pyrochlore, Gd₂TiO₅. The sequential reaction pathway is as follows [7]:
- Formation of Pyrochlore Intermediate:
Gd₂O₃ + 2 TiO₂ → Gd₂Ti₂O₇ - Formation of Stuffed Pychlore:
Gd₂Ti₂O₇ + Gd₂O₃ → 2 Gd₂TiO₅
In the MSS route, this transformation proceeds to completion at 1300 °C within 2 hours. In contrast, the SSS route exhibits slower kinetics, leaving residual Gd₂Ti₂O₇ and Gd₂O₃ even after 24 hours at 1500 °C, as confirmed by quantitative phase analysis [7].
The following diagram illustrates the shared reaction pathway but highlights the critical divergence in efficiency and completeness between the two synthesis methods.
Performance and Product Characterization
Quantitative Comparison of Synthesis Outcomes
The fundamental differences in the synthesis mechanisms lead to directly observable disparities in the quality and characteristics of the final Gd₂TiO₅ product. The key performance metrics are summarized in the table below.
Table 2: Experimental Outcomes for Gd₂TiO₅ Synthesis [7]
| Characteristic | Solid-State Synthesis (SSS) | Molten Salt Synthesis (MSS) |
|---|---|---|
| Phase Purity (at 1300-1500°C) | Contains trace Gd₂Ti₂O₇ and Gd₂O₃ impurities | Single-phase Gd₂TiO₅ |
| Particle Size | 2–3 μm, angular, irregular | ~1 μm, faceted, uniform |
| Particle Morphology | Angular, sintered agglomerates | Faceted, similar to TiO₂ precursor template |
| Reaction Kinetics | Slow (days) | Fast (hours) |
| Energy Consumption | High (higher T, longer t) | Lower (lower T, shorter t) |
The Underlying MSS Mechanism: Template Growth
The superior performance of MSS for synthesizing Gd₂TiO₅ can be attributed to its reaction mechanism. In this specific system, a template growth mechanism (a type of solution-diffusion process) is dominant [7]. This mechanism occurs because the dissolution rates of the two oxide precursors in the molten salt are significantly different.
The process can be visualized as follows:
The mechanism involves several key steps [7]:
- Precursor Immersion: The Gd₂O₃ and TiO₂ particles are suspended in the molten NaCl:KCl eutectic.
- Selective Dissolution: Gd₂O₃, having a higher solubility in the salt melt, dissolves and releases Gd³⁺ ions into the liquid medium. TiO₂, with its very low solubility, remains largely as solid particles.
- Cation Transport: The dissolved Gd³⁺ cations are transported through the liquid salt and diffuse to the surface of the less-soluble TiO₂ particles.
- Heterogeneous Reaction: The reaction between the dissolved Gd³⁺ and the solid TiO₂ template occurs at the interface, forming the product layer of Gd₂TiO₅.
- Product Formation: The reaction proceeds until the entire TiO₂ particle is consumed, resulting in a Gd₂TiO₅ particle that retains the original size and faceted morphology of the TiO₂ precursor. This explains the morphological similarity observed in SEM micrographs between the TiO₂ reagent and the final MSS product [7].
The Scientist's Toolkit: Essential Research Reagents
The experimental protocols for synthesizing Gd₂TiO₅ require specific reagents and equipment. The following table details the essential components of the "research toolkit" for these methods.
Table 3: Essential Research Reagents and Equipment for Gd₂TiO₅ Synthesis
| Item | Function / Role | Specifications / Notes |
|---|---|---|
| Gd₂O₃ (Gadolinium Oxide) | Ceramic precursor providing the Gd³⁺ cation. | Must be pre-dried at 800 °C prior to use [7]. |
| TiO₂ (Titanium Dioxide) | Ceramic precursor providing the Ti⁴⁺ cation and acting as a physical template in MSS. | Pre-dried at 180 °C. Particle size and morphology influence the final product in MSS [7]. |
| NaCl:KCl Eutectic | Molten salt solvent for MSS. Creates a liquid medium for enhanced mass transport. | Used at a 1:1 molar ratio. Lowers the effective reaction temperature. Must be removed by washing with water post-synthesis [7]. |
| Planetary Ball Mill | Equipment for homogenizing and size-reducing precursor mixtures. | Higher energy used for SSS; lower energy used for MSS to preserve template particles [7]. |
| High-Temperature Furnace | Equipment for achieving the solid-state reaction. | Must be capable of sustained operation at temperatures up to 1500 °C [7]. |
Application Context and Synthesis Selection
The choice between MSS and SSS can be guided by the intended application of the Gd₂TiO₅ material, as different applications prioritize different material properties.
- Nuclear Applications: Gd₂TiO₅ is considered for actinide waste forms and neutron absorbers in control rods due to its radiation tolerance [7] [1]. For these applications, high phase purity is critical to ensure predictable long-term chemical and physical stability under radiation. The ability of MSS to produce a single-phase material at a lower temperature gives it a distinct advantage [7].
- Phosphors and Optical Applications: Gd₂TiO₅, when doped with ions like Dy³⁺ and Eu³⁺, serves as a high-performance nanophosphor for warm white light-emitting diodes (pc-WLEDs) [11]. For such optical applications, phase purity and controlled particle morphology/morphology are essential to achieve high quantum efficiency and photostability. While the cited phosphor study used a sol-gel method, the principles of needing pure, uniform particles align with the strengths of MSS [11].
This comparison guide demonstrates that Molten Salt Synthesis offers significant advantages over conventional Solid-State Synthesis for the production of Gd₂TiO₅ stuffed pyrochlore. The data unequivocally shows that MSS achieves superior phase purity at a lower temperature (1300 °C vs. >1500 °C) and in a fraction of the time (2 hours vs. 24 hours). The core of this efficiency lies in the template growth mechanism, where the molten salt acts as a fast transport medium for reactants, leading to a more kinetically favorable pathway. The resulting material exhibits not only higher purity but also a more uniform and controllable particle morphology. For research and industrial applications where purity, energy efficiency, and particle characteristics are paramount—such as in nuclear waste forms or advanced phosphors—MSS presents itself as a compelling and robust synthetic alternative.
Conventional Solid-State Synthesis (SSS): A Traditional Approach Solid-state synthesis is a foundational method in inorganic materials science, enabling the creation of a vast array of ceramics and minerals. As a cornerstone technique, Conventional Solid-State Synthesis (SSS) relies on the high-temperature reaction of solid precursors. This guide objectively compares SSS with the emerging Molten Salt Synthesis (MSS) method, using the synthesis of gadolinium titanate (Gd₂TiO₅) "stuffed" pyrochlore—a material of interest for nuclear applications and phosphors—as a detailed case study [7] [9] [1].
Solid-State Synthesis (SSS) is a direct, "shake and bake" ceramic method where solid powder precursors are mixed and heated to high temperatures to facilitate a chemical reaction via solid-state diffusion [15]. This approach is one of the most common and historically significant routes for preparing multinary oxide ceramics, including pyrochlores and perovskites [12]. The process is driven by the thermodynamic minimization of free energy, often leading to the most stable polymorph of a material. However, this same drive toward global equilibrium can make the synthesis of metastable phases challenging [15]. The method is valued for its conceptual simplicity and the high purity of its products, but it often demands high temperatures, prolonged reaction times, and repeated grinding and heating steps to achieve homogeneity [7] [12].
The SSS Workflow: A Step-by-Step Protocol
The synthesis of a material via SSS follows a systematic, though often labor-intensive, sequence. The protocol for synthesizing Gd₂TiO₅, as derived from experimental reports, is outlined below [7] [1].
Detailed Experimental Protocol for Gd₂TiO₅ Synthesis
- Step 1: Precursor Preparation. The starting oxides, Gd₂O₃ and TiO₂, are first dried overnight at 800°C and 180°C, respectively, to remove any adsorbed moisture [7].
- Step 2: Stoichiometric Weighing and Milling. Stoichiometric amounts of the precursors are accurately weighed out to achieve the desired Gd₂TiO₅ composition. The powder mixture is then subjected to planetary ball milling. A typical procedure involves using isopropanol as a carrier fluid and milling at 500 rpm for 15 minutes to ensure intimate mixing and size reduction of the reactant particles [7].
- Step 3: Pelletization. The resulting slurry is dried and passed through a sieve (e.g., 250 μm). The powder is then uniaxially pressed into pellets (e.g., 13 mm diameter) under high pressure (e.g., 3 tonnes for 1 minute) to improve inter-particle contact and reaction kinetics [7].
- Step 4: High-Temperature Calcination. The green pellets are placed in a furnace and heated to temperatures ranging from 700°C to 1500°C. A representative synthesis involves a final heat treatment at 1300°C for 24 hours, with controlled heating and cooling rates (e.g., 5°C/min) [7] [1]. Multiple such cycles with intermediate grinding are often required to obtain a phase-pure product [12].
The following diagram illustrates this multi-step workflow and the key transformations occurring at each stage.
The Scientist's Toolkit: Key Research Reagent Solutions
The following table details the essential materials and equipment required to perform a conventional solid-state synthesis of Gd₂TiO₅.
Table 1: Essential Research Reagents and Equipment for SSS of Gd₂TiO₅
| Item | Function in the Synthesis Protocol |
|---|---|
| Gd₂O₃ (Gadolinium Oxide) | Serves as the gadolinium precursor. Must be dried to ensure stoichiometric accuracy [7]. |
| TiO₂ (Titanium Dioxide) | Serves as the titanium precursor. Anatase or rutile phases can be used [7]. |
| Planetary Ball Mill | Equipment used for the intimate mixing and size reduction of precursor powders to enhance reactivity [7]. |
| Uniaxial Press | Equipment used to compress mixed powders into dense pellets to maximize inter-particle contact during heating [7]. |
| High-Temperature Furnace | Essential equipment capable of sustaining temperatures up to 1500°C for extended durations to drive solid-state diffusion [7]. |
SSS vs. MSS: A Quantitative Comparison Using Gd₂TiO₅
To objectively evaluate performance, the table below compares key experimental data for the synthesis of Gd₂TiO₅ via SSS and the alternative Molten Salt Synthesis (MSS) method [7] [1].
Table 2: Performance Comparison: SSS vs. Molten Salt Synthesis (MSS) for Gd₂TiO₅
| Parameter | Conventional Solid-State (SSS) | Molten Salt Synthesis (MSS) |
|---|---|---|
| Synthesis Temperature | 1300°C - 1500°C | 1300°C |
| Reaction Time | 24 hours per cycle | 2 hours |
| Phase Purity Outcome | Trace impurities (Gd₂Ti₂O₇, Gd₂O₃) often persist even at 1500°C [7]. | Single-phase Gd₂TiO₅ achieved at 1300°C [7]. |
| Particle Morphology | Larger, angular particles (2-3 μm) resulting from solid-state diffusion and grain growth [7]. | Smaller, faceted particles (~1 μm) templated on the TiO₂ precursor [7]. |
| Primary Mechanism | Solid-state diffusion [12]. | Solution-diffusion or "template growth" in a liquid flux [7] [12]. |
Critical Analysis and Discussion
The comparative data reveals the distinct advantages and limitations of the conventional solid-state method.
- Efficiency and Phase Purity: The data shows that MSS offers superior efficiency, achieving a single-phase product in a fraction of the time required by SSS [7]. The SSS process, even after 24 hours at 1500°C, can leave trace amounts of intermediate phases like Gd₂Ti₂O₇ and unreacted Gd₂O₃ [7]. This highlights a key challenge in SSS: achieving complete homogeneity and reaction completion through solid-state diffusion alone is often kinetically hindered.
- Particle Characteristics: The particle morphology also differs significantly. SSS typically produces larger, more angular particles (2-3 μm) due to high-temperature coarsening and grain growth [7]. In contrast, MSS can produce smaller, more uniform particles whose shape is inherited from one of the precursors via a "template growth" mechanism [7]. This makes MSS advantageous for applications where high surface area or specific particle morphology is desired.
- Brother Context and Applicability: The persistence of the pyrochlore phase Gd₂Ti₂O₇ as a kinetic product in the synthesis of the "stuffed" pyrochlore Gd₂TiO₅ is a crucial observation [7] [1]. This illustrates a common phenomenon in SSS, where the reaction pathway proceeds through intermediate phases before reaching the thermodynamic equilibrium product. Understanding this pathway is essential for optimizing synthesis parameters, not just for this material, but for complex ceramic systems in general [15].
Conventional Solid-State Synthesis remains a vital and widely used technique in the materials scientist's toolkit, prized for its directness and the high-purity products it can yield. However, as the direct comparison with Molten Salt Synthesis for Gd₂TiO₅ demonstrates, SSS is often characterized by higher energy consumption, longer processing times, and challenges in achieving perfect phase purity and controlled particle size. The choice of synthesis method ultimately depends on the target material's required properties, its thermodynamic stability, and the application's specific needs for phase purity, particle size, and morphology. For the synthesis of refractory "stuffed" pyrochlores like Gd₂TiO₅, MSS presents a compelling, more efficient alternative to the traditional SSS approach.
The synthesis of advanced ceramic materials, such as the 'stuffed' pyrochlore Gd₂TiO₅, is critically dependent on precise control over process parameters including temperature, time, and atmospheric conditions. These parameters directly influence phase purity, morphological characteristics, and functional performance in specialized applications such as nuclear waste forms and neutron absorbers [7]. Within materials science research, two prominent synthesis pathways—conventional solid-state synthesis (SSS) and molten salt synthesis (MSS)—offer distinct approaches to parameter control, leading to significant differences in resultant material properties [7] [13].
This comparison guide objectively evaluates these competing methodologies by examining experimental data on the synthesis of Gd₂TiO₅, providing researchers with a systematic framework for selecting appropriate synthesis techniques based on critical process parameters. The analysis presented herein is situated within a broader thesis investigating the relative advantages and limitations of molten salt versus solid-state synthesis routes for refractory ceramic materials.
Methodology Comparison: Molten Salt vs. Solid-State Synthesis
Fundamental Process Mechanisms
The fundamental mechanisms underlying solid-state and molten salt synthesis differ significantly, leading to variations in efficiency, product quality, and microstructural control.
Solid-State Synthesis (SSS) relies on direct diffusion and reaction between solid oxide precursors at elevated temperatures. This method requires repeated milling and calcination steps at high temperatures for extended durations to enhance solid-state diffusion necessary for single-phase product formation [7]. The process is inherently limited by solid-state diffusion kinetics, often resulting in incomplete reactions, large particle sizes, and potential loss of volatile constituents at high temperatures [7].
Molten Salt Synthesis (MSS) utilizes a salt or eutectic salt mixture that melts at moderate temperatures to create a liquid reaction medium. This liquid phase significantly enhances ion mobility and diffusion rates between reactant species [7]. Two primary mechanisms govern MSS:
- Solution-Precipitation: Occurs when reactant dissolution rates are comparable, leading to simultaneous dissolution into the molten salt followed by product precipitation once supersaturation is achieved [7].
- Template Growth (Solution-Diffusion): Functions when one reactant exhibits substantially higher solubility than the other. The more soluble reactant dissolves and diffuses to the surface of the less soluble reactant, where product formation occurs [7]. This mechanism often preserves the morphology of the less soluble reactant template.
Experimental Protocols for Gd₂TiO₅ Synthesis
Solid-State Synthesis Protocol
For conventional solid-state synthesis of Gd₂TiO₅, the following methodology was employed [7]:
- Precursor Preparation: Gd₂O₃ and TiO₂ reagents were dried at 800°C and 180°C, respectively, overnight.
- Stoichiometric Batching: Precursors were batched to 10 g total mass in stoichiometric ratios for Gd₂TiO₅ formation.
- Milling: The mixture was planetary ball milled at 500 rpm for 15 minutes using isopropanol as a carrier fluid.
- Drying and Sieving: The resulting slurry was dried at 80°C overnight and passed through a 250 μm sieve.
- Pelletization: 0.5 g samples were uniaxially pressed into 13 mm pellets at 3 tonnes applied pressure for 1 minute.
- Thermal Treatment: Green pellets were reacted at temperatures ranging from 700-1500°C in 100°C intervals for 24 hours at each temperature with heating and cooling rates of 5°C/min.
Molten Salt Synthesis Protocol
For molten salt synthesis of Gd₂TiO₅, the following methodology was applied [7]:
- Precursor Preparation: Gd₂O₃ and TiO₂ reagents were dried at 800°C and 180°C, respectively, overnight.
- Salt Addition: An equimolar eutectic mixture of NaCl:KCl (1:1 mol ratio) was added at a 7:1 molar ratio of molten salt to ceramic precursor.
- Milling: The salt and precursor mixture was milled together at 250 rpm for 5 minutes using cyclohexane as the carrier fluid.
- Pelletization: Green pellets were produced using the same method as for SSS.
- Thermal Treatment: Pellets were reacted at temperatures ranging from 700-1500°C in 100°C intervals for 2 hours with heating and cooling rates of 5°C/min.
- Salt Removal: The product was washed with ultrapure water to remove the molten salt medium, and the powder was collected via vacuum filtration.
Table 1: Key Process Parameter Comparison Between Synthesis Methods
| Process Parameter | Solid-State Synthesis | Molten Salt Synthesis |
|---|---|---|
| Reaction Temperature | 700-1500°C | 700-1500°C |
| Reaction Time | 24 hours at each temperature | 2 hours at each temperature |
| Atmosphere | Ambient (not specified) | Ambient (not specified) |
| Heating/Cooling Rate | 5°C/min | 5°C/min |
| Additional Steps | Repeated milling and heating cycles | Salt removal by washing |
| Mechanism | Solid-state diffusion | Template growth/solution-precipitation |
Comparative Performance Analysis
Phase Formation and Purity
X-ray diffraction analysis reveals significant differences in phase evolution and purity between the two synthesis methods across the temperature range studied [7].
Molten Salt Synthesis demonstrated rapid phase formation with Gd₂TiO₅ and Gd₂Ti₂O₇ pyrochlore observed as early as 800-900°C, though with significant residual Gd₂O₃. By 1100°C, Gd₂TiO₅ became the predominant phase with diminished Gd₂Ti₂O₇ and Gd₂O₃ reflections. Critically, single-phase Gd₂TiO₅ was achieved at 1300°C after only 2 hours of reaction time [7].
Solid-State Synthesis showed initial formation of Gd₂TiO₅ at 900°C (compared to 800°C for MSS), with Gd₂TiO₅ becoming the major phase at 1000°C. However, even at 1500°C with 24-hour reaction time, trace amounts of Gd₂Ti₂O₇ and Gd₂O₃ remained present, as evidenced by the persistent (222) reflection of Gd₂O₃ at 2θ = 29° and (111) reflection of Gd₂Ti₂O₇ at 2θ = 15° [7]. This indicates that additional heat treatments would be necessary to generate a single-phase product via SSS.
Table 2: Phase Purity Comparison at Critical Temperatures
| Synthesis Method | Temperature | Time | Phase Composition | Key Observations |
|---|---|---|---|---|
| Molten Salt Synthesis | 800-900°C | 2 hours | Gd₂TiO₅, Gd₂Ti₂O₇, Gd₂O₃ | Initial phase formation |
| 1100°C | 2 hours | Predominantly Gd₂TiO₅ | Minor impurities present | |
| 1300°C | 2 hours | Single-phase Gd₂TiO₅ | No detectable impurities | |
| Solid-State Synthesis | 900°C | 24 hours | Gd₂TiO₅, Gd₂Ti₂O₇, Gd₂O₃ | Initial phase formation |
| 1000°C | 24 hours | Major phase Gd₂TiO₅ | Significant impurities | |
| 1500°C | 24 hours | Gd₂TiO₅ with traces of Gd₂Ti₂O₇ and Gd₂O₃ | Incomplete reaction |
Microstructural Development
Scanning electron microscopy analysis reveals profound differences in particle morphology and size distribution between the two synthesis methods [7].
MSS-derived Gd₂TiO₅ particles exhibited a faceted morphology with an average size of approximately 1 μm, strikingly similar in both morphology and dimensions to the original TiO₂ reagent particles. This morphological correspondence strongly indicates a template growth mechanism, where the TiO₂ particles serve as structural templates upon which the product forms [7].
SSS-derived Gd₂TiO₅ particles were significantly larger (2-3 μm) with more angular morphology, reflecting the process of conventional solid-state diffusion and grain growth at elevated temperatures. The larger particle size in SSS materials is attributable to extended exposure to high temperatures during the lengthier synthesis process [7].
Alternative Synthesis Approaches
The "firing salts method" (FSM) represents a related synthesis approach that has demonstrated efficacy for Gd₂TiO₅ formation. This method successfully produced orthorhombic Gd₂TiO₅ at 1200°C with just 2 hours of reaction time, resulting in particles ranging between 50-500 nm in size [8]. This further confirms the general advantage of salt-assisted synthesis methods in reducing reaction temperatures and times while controlling particle size characteristics.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents and Materials for Gd₂TiO₅ Synthesis
| Reagent/Material | Function | Specifications | Application in Synthesis |
|---|---|---|---|
| Gadolinium Oxide (Gd₂O₃) | Ceramic precursor source of Gd cations | Dried at 800°C overnight | Primary reactant in both MSS and SSS |
| Titanium Dioxide (TiO₂) | Ceramic precursor source of Ti cations | Dried at 180°C overnight | Primary reactant in both MSS and SSS |
| Sodium Chloride (NaCl) | Molten salt medium component | Eutectic mixture (1:1 mol with KCl) | Creates liquid reaction environment in MSS |
| Potassium Chloride (KCl) | Molten salt medium component | Eutectic mixture (1:1 mol with NaCl) | Lowers melting point of salt medium in MSS |
| Isopropanol | Milling carrier fluid | Laboratory grade | Prevents overheating during milling process |
| Cyclohexane | Milling carrier fluid | Laboratory grade | Alternative carrier for salt-containing mixtures |
Process Workflow Visualization
Synthesis Pathway Comparison: This workflow illustrates the procedural differences between Molten Salt Synthesis (MSS) and Solid-State Synthesis (SSS) routes for Gd₂TiO₅ formation, highlighting the more efficient pathway achieved through MSS.
Parameter Optimization Guidelines
Temperature Optimization
Based on experimental results, MSS achieves single-phase Gd₂TiO₅ at 1300°C, while SSS fails to achieve complete phase purity even at 1500°C [7]. The significantly lower temperature requirement for MSS represents substantial energy savings and reduced operational costs. The temperature threshold for initial phase formation is also approximately 100°C lower in MSS (800°C) compared to SSS (900°C) [7].
Time Parameter Optimization
The time efficiency advantage of MSS is dramatic, with complete reaction achieved in just 2 hours compared to 24 hours or more for SSS [7]. When considering potential multiple cycles needed for SSS to achieve comparable phase purity, the time differential becomes even more significant. This time reduction directly translates to higher laboratory throughput and faster research cycles.
Atmosphere Considerations
While the cited studies do not explicitly detail atmospheric control, the successful implementation of both methods under ambient conditions suggests that atmospheric control may not be a critical parameter for Gd₂TiO₅ synthesis. However, for moisture-sensitive precursors or specialized doping strategies, controlled atmospheres may become necessary.
The comparative analysis presented in this guide demonstrates clear advantages of molten salt synthesis over conventional solid-state methods for the preparation of Gd₂TiO₅ stuffed pyrochlore materials. MSS achieves superior phase purity at lower temperatures (1300°C vs. >1500°C) and significantly reduced reaction times (2 hours vs. 24+ hours) while producing more uniform, smaller particles with controlled morphology through a template growth mechanism [7].
These performance advantages position MSS as a more efficient, economically viable, and environmentally sustainable synthesis route for refractory ceramic materials, particularly relevant for nuclear applications where phase purity and microstructural control directly impact material performance. The critical parameter optimization detailed in this guide provides researchers with a validated framework for selecting and implementing appropriate synthesis methodologies based on specific research objectives and material requirements.
The synthesis of advanced ceramic materials like gadolinium titanate (Gd₂TiO₅) extends beyond the initial reaction to encompass critical downstream processes that ultimately determine the material's phase purity, morphology, and applicability. As a "stuffed" pyrochlore structure, Gd₂TiO₅ has garnered significant interest for nuclear applications, including actinide waste forms and neutron absorbers, where precise control over its structural properties is paramount [7]. This comparison guide objectively examines the post-synthesis processing protocols for Gd₂TiO₅ prepared via two distinct synthetic pathways: molten salt synthesis (MSS) and conventional solid-state synthesis (SSS). By analyzing the washing, drying, and pelletizing requirements of each method alongside their final outcomes, this guide provides researchers with a practical framework for selecting and optimizing their synthesis strategy.
Molten Salt Synthesis (MSS) Protocol
The MSS route utilizes a low-melting-point salt medium to enhance ion diffusion and accelerate reaction kinetics at lower temperatures [7].
- Synthesis Procedure: Precursor powders (Gd₂O₃ and TiO₂) are mixed with an equimolar eutectic flux of NaCl:KCl. The salt-to-precursor molar ratio is maintained at 7:1. The mixture is subjected to low-energy planetary ball milling (250 rpm for 5 minutes) using cyclohexane as a carrier fluid to minimize pre-synthesis size reduction. The resulting mixture is uniaxially pressed into pellets (3 tonnes for 1 minute) and reacted at temperatures between 700–1500 °C for a short duration of 2 hours [7].
- Critical Post-Synthesis Washing: After the high-temperature reaction, the cooled product contains the soluble salt flux. To isolate the pure ceramic powder, the pellet is crushed and washed repeatedly with ultrapure water to dissolve and remove the NaCl/KCl salts. The final powder is collected via vacuum filtration [7].
- Drying and Pelletizing: The washed filter cake is typically dried overnight at 80°C. The resulting pure, fine powder can then be pressed into new pellets for further characterization or sintering [7].
Solid-State Synthesis (SSS) Protocol
The SSS method relies on direct, high-temperature reaction of solid oxide precursors, requiring more energy and longer processing times.
- Synthesis Procedure: Stoichiometric Gd₂O₃ and TiO₂ are mixed and subjected to high-energy planetary ball milling (500 rpm for 15 minutes) in isopropanol to achieve intimate mixing and particle size reduction. The dried powder is pressed into pellets and reacted at 700–1500 °C for significantly longer periods, typically 24 hours per cycle, often with intermediate grinding and re-pellettizing to improve homogeneity [7] [9].
- Post-Synthesis Processing: A key differentiator is that the SSS product does not require a washing step, as no flux media is used. The reacted pellets are ground into powder for characterization. For the photoluminescence study, powders after the final calcination were used directly without further compaction [7] [9].
The following workflow diagram visualizes the procedural divergence between these two methods, particularly highlighting the post-synthesis stages.
Performance and Outcome Comparison
The choice of synthesis method directly influences the efficiency, phase purity, and morphology of the final Gd₂TiO₅ product, with significant implications for downstream applications.
Table 1: Quantitative Comparison of Synthesis Outcomes for Gd₂TiO₅
| Parameter | Molten Salt Synthesis (MSS) | Conventional Solid-State Synthesis (SSS) |
|---|---|---|
| Reaction Temperature & Time | 1300 °C for 2 hours [7] | 1500 °C for 24 hours (trace impurities remain) [7] |
| Phase Purity | Single-phase Gd₂TiO₅ achieved at 1300°C [7] | Trace amounts of Gd₂Ti₂O₇ and Gd₂O₃ even at 1500°C [7] |
| Particle Morphology | Faceted particles, ~1 μm, resembling TiO₂ precursor template [7] | Larger, angular particles, 2-3 μm [7] |
| Primary Mechanism | Template growth (Solution-Diffusion) [7] | Solid-state diffusion [7] |
| Key Post-Synthesis Step | Washing and filtration to remove salt flux [7] | No washing required [7] |
The data reveals a clear trade-off. The MSS method, while introducing an extra washing step, offers superior kinetics and phase purity. The liquid medium facilitates rapid diffusion, allowing the formation of single-phase Gd₂TiO₅ at a lower temperature and in a fraction of the time (2 hours vs. 24+ hours) required by SSS [7]. The "template growth" mechanism in MSS, where the TiO₂ particle acts as a seed, results in a finer and more uniform morphology compared to the larger, sintered aggregates typical of SSS [7]. In contrast, SSS struggles with kinetic limitations, often retaining impurity phases like Gd₂Ti₂O₇ and unreacted Gd₂O₃ even after prolonged high-temperature treatment [7].
The Scientist's Toolkit: Essential Research Reagents
Successful synthesis and processing of Gd₂TiO₅ depend on the use of specific, high-purity reagents and equipment.
Table 2: Essential Reagents and Equipment for Gd₂TiO₅ Synthesis
| Item | Function & Specification | Synthesis Method |
|---|---|---|
| Gd₂O₃ (99.9%) | High-purity rare earth oxide precursor [7] [9]. | MSS & SSS |
| TiO₂ (99.0%) | Titanium source; particle morphology templates MSS product [7] [9]. | MSS & SSS |
| NaCl:KCl Eutectic | Molten salt flux (1:1 mol ratio); creates liquid reaction medium [7]. | MSS |
| Planetary Ball Mill | For homogenizing reactants (SSS: high-energy; MSS: low-energy) [7]. | MSS & SSS |
| Uniaxial Press | Forms green pellets (e.g., 3 tonnes for 1 min) for high-temperature reaction [7]. | MSS & SSS |
| Ultrapure Water | Critical for post-synthesis washing to remove soluble salt flux [7]. | MSS |
| Vacuum Filtration Setup | For efficient solid-liquid separation after the washing step [7]. | MSS |
The post-synthesis processing workflow for Gd₂TiO₅ is intrinsically tied to the chosen synthesis method. Molten Salt Synthesis introduces a mandatory washing and filtration step but rewards the researcher with highly crystalline, single-phase material at lower temperatures and significantly shorter reaction times, offering a compelling advantage for efficiency and phase purity. Conventional Solid-State Synthesis avoids the washing step but demands higher temperatures, prolonged heating, and often yields a product with residual impurities and less controlled morphology. The decision between these methods ultimately hinges on the researcher's priority: MSS for speed and phase purity with added processing, or SSS for a simpler, albeit more energy-intensive and time-consuming, direct reaction.
Overcoming Synthesis Challenges: Impurities, Kinetics, and Scalability
Common Impurities and Intermediate Phase Formation
The synthesis of single-phase gadolinium titanate (Gd₂TiO₅) is of significant interest for advanced technological applications, particularly in the nuclear industry where it serves as a potential matrix for actinide disposition and in control rods due to its favorable neutron absorption characteristics [7]. The pathway to achieving a pure, single-phase material is complicated by the formation of specific intermediate phases and persistent impurities, which are heavily influenced by the chosen synthesis methodology. This guide provides a comparative analysis of two prominent synthesis routes—conventional solid-state synthesis (SSS) and molten salt synthesis (MSS)—focusing on the identity and persistence of intermediate phases and impurities, supported by experimental data and detailed protocols.
Synthesis Methods and Experimental Protocols
The formation of Gd₂TiO₅ is highly dependent on the synthesis technique, which directly impacts the reaction kinetics, diffusion pathways, and ultimate phase purity.
Conventional Solid-State Synthesis (SSS) Protocol
The conventional solid-state method is a widely used ceramic processing route that relies on high temperatures and long durations to facilitate solid-state diffusion between reactant oxides [7].
- Reagents: Gd₂O₃ and TiO₂ are used as precursor materials.
- Pre-treatment: Precursors are dried overnight at 800 °C and 180 °C, respectively, to remove adsorbed moisture.
- Milling: Stoichiometric amounts of the reagents are batched and subjected to planetary ball milling at 500 rpm for 15 minutes, using isopropanol as a carrier fluid to ensure homogeneous mixing.
- Pelletization: The resulting powder slurry is dried and sieved through a 250 μm mesh. The powder is then uniaxially pressed into pellets under a load of 3 tonnes for 1 minute.
- Reaction: The green pellets are reacted at temperatures ranging from 700 °C to 1500 °C, typically for extended durations of 24 hours per cycle, with a heating and cooling rate of 5 °C/min [7].
Molten Salt Synthesis (MSS) Protocol
Molten salt synthesis utilizes a eutectic salt mixture as a liquid reaction medium to enhance diffusion and reduce synthesis time and temperature [7].
- Reagents: Gd₂O₃ and TiO₂ (dried as above).
- Salt Flux: An equimolar eutectic mixture of NaCl and KCl is used. A 7:1 molar ratio of molten salt to ceramic precursor is employed.
- Milling: The salt and precursor mixture are co-milled at a lower energy input (250 rpm for 5 minutes) using cyclohexane as the carrier fluid to minimize excessive size reduction.
- Pelletization: Powder is pressed into pellets as described for SSS.
- Reaction: Pellets are reacted at temperatures from 700 °C to 1500 °C for a significantly shorter duration of 2 hours at the target temperature, with the same 5 °C/min ramp rate.
- Post-processing: The reacted product is washed with ultrapure water to dissolve and remove the salt flux, and the resulting powder is collected via vacuum filtration [7].
Comparative Analysis of Phase Formation and Impurities
The phase progression and persistence of impurities differ markedly between the two synthesis methods. The primary intermediate phase identified in both routes is the pyrochlore Gd₂Ti₂O₇, which forms as a kinetic product before transforming into the orthorhombic Gd₂TiO₅ phase [7].
Table 1: Phase Progression and Key Impurities in Gd₂TiO₅ Synthesis
| Synthesis Method | Temperature Range for Gd₂TiO₅ Formation | Key Intermediate Phase | Persistent Impurities at High Temperature (e.g., 1500 °C) |
|---|---|---|---|
| Solid-State Synthesis (SSS) | First observed at 900 °C; major phase at 1000 °C [7] | Gd₂Ti₂O₇ [7] | Trace Gd₂Ti₂O₇ and unreacted Gd₂O₃ [7] |
| Molten Salt Synthesis (MSS) | Predominant at 1100 °C; single-phase at 1300 °C [7] | Gd₂Ti₂O₇ [7] | None reported; single-phase Gd₂TiO₅ achieved at 1300 °C [7] |
Table 2: Quantitative Comparison of Synthesis Parameters and Outcomes
| Parameter | Solid-State Synthesis (SSS) | Molten Salt Synthesis (MSS) |
|---|---|---|
| Reaction Time | 24 hours per cycle [7] | 2 hours [7] |
| Temperature for Single Phase | >1500 °C (single phase not achieved) [7] | 1300 °C [7] |
| Particle Morphology | Larger (2-3 μm), angular particles [7] | Smaller (~1 μm), faceted particles, templated from TiO₂ precursor [7] |
| Proposed Mechanism | Solid-state diffusion [7] | Template growth (solution-diffusion) [7] |
The data demonstrates that MSS offers a more efficient pathway to phase-pure Gd₂TiO₅, achieving the target compound at a lower thermal budget and without the persistent impurities that characterize the SSS route.
Mechanism of Molten Salt Synthesis
The superior performance of MSS is attributed to a template growth mechanism (a type of solution-diffusion process). In this mechanism, the reactants have dissimilar dissolution rates in the molten salt medium. The TiO₂ particles, which have very low solubility in alkali chlorides, act as a template. The Gd₂O₃, which is more soluble, dissolves into the molten salt, diffuses to the surface of the TiO₂ particles, and reacts to form the product phase, effectively preserving the morphology of the TiO₂ template [7]. This process is illustrated below.
The Scientist's Toolkit: Key Research Reagents and Materials
The following table details essential materials and their functions in the synthesis of Gd₂TiO₅, as derived from the experimental protocols.
Table 3: Essential Reagents and Materials for Gd₂TiO₅ Synthesis
| Reagent/Material | Function in Synthesis | Key Specifications & Pre-Treatment |
|---|---|---|
| Gadolinium Oxide (Gd₂O₃) | Primary gadolinium source precursor. | Purity ≥ 99.9%. Dried overnight at 800 °C to remove moisture and oxycarbonates [7] [9]. |
| Titanium Dioxide (TiO₂) | Primary titanium source precursor. | Purity ≥ 99.9%. Dried overnight at 180 °C [7] [9]. Acts as a template in MSS due to low solubility [7]. |
| Sodium Chloride (NaCl) | Component of eutectic salt flux (MSS). | Forms a low-melting-point eutectic with KCl. Must be dried (e.g., at 110 °C) before use [7]. |
| Potassium Chloride (KCl) | Component of eutectic salt flux (MSS). | Forms a low-melting-point eutectic with NaCl. Must be dried (e.g., at 110 °C) before use [7]. |
| Isopropanol / Cyclohexane | Carrier fluid for milling. | Facilitates homogeneous mixing during powder milling and prevents excessive dust formation [7]. |
The formation of impurities and intermediate phases in the synthesis of Gd₂TiO₅ is a critical consideration for researchers. The evidence clearly indicates that the pyrochlore phase Gd₂Ti₂O₇ is a common kinetic intermediate, but its persistence and the concomitant presence of unreacted Gd₂O₃ are far more problematic in conventional solid-state synthesis. Molten salt synthesis, operating through an efficient template growth mechanism, presents a superior alternative. It significantly reduces reaction time and temperature and effectively eliminates persistent impurities, yielding single-phase Gd₂TiO₅ where solid-state synthesis struggles. This comparison provides a definitive guide for researchers in selecting a synthesis method tailored to their requirements for phase purity, processing time, and particle morphology.
Strategies for Achieving Single-Phase Purity
In the field of advanced ceramic materials, the pursuit of single-phase purity is a fundamental objective, directly influencing the physical, thermal, and optical properties of the final product. This guide objectively compares the performance of conventional solid-state synthesis against molten salt methods for synthesizing gadolinium titanate (Gd₂TiO₅), a material of significant interest for nuclear applications and phosphor matrices. The context is framed within a broader thesis on synthesis route optimization, providing researchers with experimental data and protocols to inform their methodological choices. Gd₂TiO₅ belongs to a class of 'stuffed' pyrochlores and orthorhombic structures, whose performance in applications such as neutron absorption or luminescent hosts is critically dependent on phase purity and controlled microstructure [9] [1].
The synthesis of Gd₂TiO₅ presents specific challenges due to the formation of intermediate phases and the refractory nature of the constituent oxides. Achieving single-phase Gd₂TiO₅ requires careful control over reaction kinetics, temperature, and time to bypass the formation of the common pyrochlore phase, Gd₂Ti₂O₇, which often appears as a kinetic product. The following sections detail the core methodologies, with their experimental protocols and a direct performance comparison.
Conventional Solid-State Synthesis
Experimental Protocol: The conventional solid-state method is a widely used ceramic processing route. The detailed protocol for synthesizing Gd₂TiO₅ is as follows [9]:
- Precursor Preparation: Start with binary constituent oxides, Gd₂O₃ and TiO₂. Subject both powders to an overnight heat treatment at 800 °C to remove adsorbed moisture and oxycarbonate species.
- Stoichiometric Weighing: Accurately weigh the heat-treated powders in a stoichiometric molar ratio corresponding to the Gd₂TiO₅ composition.
- Mixing and Grinding: Mechanically mix and grind the powders using an agate mortar and pestle or a ball mill to achieve a homogeneous mixture and increase surface area for the solid-state reaction.
- Calcination: Subject the mixed powders to a high-temperature calcination process. The protocol involves a multi-step heating profile. The mixture is first heated to 1100 °C for 12 hours. After intermediate grinding to ensure homogeneity, a second heat treatment is performed at 1200 °C for 24 hours. Finally, the powder is pressed into pellets and given a final heat treatment at 1300 °C for 24 hours to achieve a single-phase product.
- Characterization: The final product is characterized by powder X-ray diffraction (XRD) to confirm phase purity and crystal structure.
Molten Salt Synthesis (MSS)
Experimental Protocol: Molten Salt Synthesis offers a lower-temperature, faster alternative by using a salt medium to enhance ion diffusion. The protocol for Gd₂TiO₅ is as follows [1]:
- Precursor and Salt Selection: Begin with the same precursor oxides, Gd₂O₃ and TiO₂. Select an appropriate salt or salt mixture; chlorides are commonly used (e.g., NaCl, KCl, or their eutectic mixtures).
- Mixing with Salt: Combine the precursor oxides with the salt in a typical weight ratio of 1:1 to 2:1 (oxide reactants to salt). Thoroughly mix the powders to ensure uniform distribution.
- Heating and Reaction: Heat the mixture in an alumina crucible to a temperature above the melting point of the salt, typically between 1200 °C. The molten salt acts as a solvent, facilitating the dissolution and reaction of the oxide precursors.
- Dwell Time: Maintain the temperature for a significantly shorter duration than solid-state synthesis; 2 hours at 1300 °C has been shown to produce single-phase Gd₂TiO₅.
- Cooling and Washing: After the reaction, cool the crucible to room temperature. The solidified salt mass is then crushed and repeatedly washed with hot deionized water or dilute acid to remove the water-soluble salt, leaving behind the synthesized Gd₂TiO₅ powder.
- Characterization: The final powder is characterized by XRD to confirm phase formation and purity.
Table 1: Quantitative Comparison of Synthesis Methods for Gd₂TiO₅
| Parameter | Conventional Solid-State | Molten Salt Synthesis |
|---|---|---|
| Typical Temperature | 1300 °C | 1200 - 1300 °C |
| Typical Time | 60 hours (including multiple steps) | 2 hours |
| Key Intermediate | Gd₂Ti₂O₇ (pyrochlore) | Gd₂Ti₂O₇ (pyrochlore) |
| Reaction Mechanism | Direct solid-state diffusion | Solution-precipitation, template growth |
| Particle Morphology | Irregular, larger aggregates | Controlled, can be plate-like/sheet-like |
| Primary Advantage | High yield, no post-washing | Faster kinetics, lower temperature, controlled morphology |
Experimental Data and Performance Metrics
The effectiveness of a synthesis strategy is ultimately judged by the quality and properties of the final material. The following data, drawn from experimental studies, provides a quantitative basis for comparing the outcomes of different synthesis routes for Gd₂TiO₅ and related compounds.
Phase Purity and Structural Analysis
X-ray diffraction (XRD) is the primary tool for verifying phase purity. Studies confirm that the molten salt method can achieve single-phase, orthorhombic Gd₂TiO₅ at 1300 °C in just 2 hours, bypassing the kinetic stabilization of the Gd₂Ti₂O₇ pyrochlore phase through a template growth mechanism [1]. Similarly, the "firing salts method," a variant of MSS, has successfully synthesized orthorhombic Gd₂TiO₅ at 1200 °C in 2 hours, with Rietveld analysis confirming the structure and electron microscopy showing a particle size distribution between 50–500 nm [8]. In contrast, conventional solid-state synthesis requires prolonged heating at similar temperatures to achieve the same phase-pure outcome.
Functional Performance in Applications
The synthesis route can profoundly impact functional properties. In luminescent applications, phase purity is critical for performance. Gd₂TiO₅ doped with Eu³⁺ (GTOE), synthesized via solid-state reaction, has demonstrated exceptional credentials as a red phosphor. Key performance metrics include:
- High Dopant Solubility: No fluorescence concentration quenching up to 10 mol% Eu³⁺ doping.
- High Color Purity: Approximately 99%, which is crucial for display and lighting applications.
- Improved Photoluminescence Quantum Yield (PLQY): An absolute PLQY of ~14%, indicating efficient light emission [9].
Table 2: Impact of Synthesis on Material Properties and Applications
| Property/Metric | Conventional Solid-State (Gd₂TiO₅:Eu³⁺) | Molten Salt Synthesis (General) |
|---|---|---|
| Photoluminescence | High color purity (~99%) and PLQY (~14%) demonstrated [9] | (Data specific to luminescence of MSS-made Gd₂TiO₅ not available in search results) |
| Particle Morphology | Not specifically reported, typically irregular | Plate-like/sheet morphology achievable [8] [14] |
| Particle Size Control | Limited control, larger aggregates | Fine control, nanosized particles (50-500 nm) possible [8] |
| Band Gap Engineering | Indirectly via doping; minimal reduction in band gap with Eu doping observed [9] | Synthesis temperature can influence band gap, as seen in analogous Bi-based titanates [14] |
The Scientist's Toolkit: Essential Research Reagents
The following table details key reagents and materials essential for the synthesis of Gd₂TiO₅, along with their specific functions in the experimental process.
Table 3: Key Research Reagents for Gd₂TiO₅ Synthesis
| Reagent/Material | Function in Synthesis | Purity & Sourcing Considerations |
|---|---|---|
| Gadolinium Oxide (Gd₂O₃) | Primary source of Gd³⁺ cations. | High purity (≥99.9%) from suppliers like Indian Rare Earths Ltd. is used to minimize impurities [9]. |
| Titanium Dioxide (TiO₂) | Primary source of Ti⁴⁺ cations. | High purity (≥99.0%) from suppliers like Sigma-Aldrich is typical [9]. |
| Europium Oxide (Eu₂O₃) | Dopant ion for photoluminescence studies, substituting for Gd³⁺. | Purity of ≥99.0% is standard [9]. |
| Sodium Chloride (NaCl)/Potassium Chloride (KCl) | Molten salt medium. Lowers synthesis temperature, enhances diffusion, and can control particle morphology. | Common laboratory salts. Often used in a eutectic mixture to achieve a lower melting point [1] [14]. |
| Alumina Crucible | Container for high-temperature reactions. | Must be chemically inert to the reactants and molten salt at operating temperatures (up to 1300°C). |
Workflow and Decision Pathways
The journey to phase-pure Gd₂TiO₅ involves a series of critical decisions, from method selection to final characterization. The following diagram maps this experimental workflow, highlighting the divergent paths of the two primary synthesis strategies.
Diagram 1: Experimental Workflow for Gd₂TiO₅ Synthesis
The strategic selection of a synthesis method is paramount in the pursuit of single-phase Gd₂TiO₅. Conventional solid-state synthesis remains a robust, high-yield method capable of producing high-quality material suitable for demanding applications such as efficient phosphors, as evidenced by the excellent photoluminescent properties of Eu³⁺-doped Gd₂TiO₅. However, the molten salt synthesis method presents a compelling alternative, offering significant advantages in efficiency, energy consumption, and morphological control. Its ability to achieve phase purity at comparable temperatures in a fraction of the time (2 hours vs. 60 hours) makes it a powerful tool for materials research and development [1]. The choice between these methods ultimately depends on the researcher's priorities: whether they are the supreme functional performance of the bulk material or the efficiency and microstructural control offered by advanced synthesis techniques. This comparison provides a foundational framework for making that critical decision.
Controlling Particle Morphology and Size Distribution
The synthesis of ceramic materials with well-defined particle morphology and size distribution is a critical objective in materials science, directly influencing the performance of ceramics in applications ranging from nuclear waste forms to solid-state lighting. The synthesis route plays a determining role in achieving this control. This guide provides a comparative analysis of molten salt synthesis (MSS) and conventional solid-state synthesis (SSS) for producing gadolinium titanate (Gd₂TiO₅), a refractory "stuffed" pyrochlore of significant interest. By examining experimental data and protocols, we objectively evaluate the efficacy of each method in controlling key particulate characteristics.
Synthesis Mechanisms and Morphological Outcomes
The fundamental mechanisms of MSS and SSS differ substantially, leading to distinct morphological outcomes.
Molten Salt Synthesis (MSS): Template Growth Mechanism
MSS utilizes a molten salt medium (e.g., a eutectic mixture of NaCl:KCl) to enhance ion diffusion at lower temperatures and shorter time scales compared to SSS [7]. For Gd₂TiO₅ synthesis, a template growth mechanism is observed [7]. In this process, the dissolution rates of the reactant oxides are mismatched; Gd₂O₃ dissolves rapidly into the molten salt flux, while TiO₂ particles remain largely undissolved, acting as a template. The dissolved Gd₂O³⁺ ions then diffuse through the liquid salt to the surface of the TiO₂ particles, where they react to form the Gd₂TiO₅ product. This mechanism directly results in the formation of submicron, faceted particles with an average size of approximately 1 μm, directly replicating the morphology of the original TiO₂ precursor [7].
Solid-State Synthesis (SSS): Solid-State Diffusion Mechanism
SSS relies entirely on solid-state diffusion between mixed solid precursors (Gd₂O₃ and TiO₂) [7]. This process requires repeated milling and high-temperature treatments for extended durations to achieve homogeneity and complete the reaction. The mechanism involves direct atomic diffusion across grain boundaries at points of contact between precursor particles, followed by nucleation and growth of the product phase. This process typically leads to the formation of larger, irregular, and angular particles with a typical size of 2-4 μm, which is a direct consequence of high-temperature sintering and particle coarsening [7].
Table 1: Comparison of Synthesis Mechanisms and Morphological Outcomes for Gd₂TiO₅
| Feature | Molten Salt Synthesis (MSS) | Conventional Solid-State Synthesis (SSS) |
|---|---|---|
| Primary Mechanism | Template growth/Solution-diffusion [7] | Solid-state diffusion [7] |
| Driving Force | Dissolution and diffusion in a liquid medium [7] | Atomic diffusion at points of solid particle contact [7] |
| Typical Particle Morphology | Faceted, homogeneous [7] | Angular, irregular [7] |
| Average Particle Size | ~1 μm [7] | 2-3 μm [7] |
| Size Distribution | Narrower (mimics template) | Broader |
The following diagram illustrates the key mechanistic differences between the two synthesis pathways:
Quantitative Performance Comparison
Direct experimental comparison of MSS and SSS for Gd₂TiO₅ synthesis reveals stark differences in reaction conditions, phase purity, and the resulting particle characteristics.
Table 2: Experimental Data Comparison for Gd₂TiO₅ Synthesis [7]
| Parameter | Molten Salt Synthesis (MSS) | Conventional Solid-State Synthesis (SSS) |
|---|---|---|
| Temperature for Single-Phase Product | 1300 °C | >1500 °C (trace impurities remain) |
| Time at Temperature | 2 hours | 24 hours |
| Phase Purity at 1300°C | Single-phase Gd₂TiO₅ | Gd₂TiO₅ with Gd₂Ti₂O₇ and Gd₂O₃ impurities |
| Particle Morphology | Faceted | Angular |
| Particle Size | ~1 μm | 2-3 μm |
| Key Advantage | Lower temperature, shorter time, high phase purity, morphology control | Simplicity of setup |
Detailed Experimental Protocols
To achieve the reported results, specific experimental protocols must be followed. The following reagent table outlines the essential materials for these syntheses.
Table 3: Key Research Reagent Solutions for Gd₂TiO₅ Synthesis
| Reagent/Material | Function in Synthesis | Example Source & Purity |
|---|---|---|
| Gd₂O₃ | Gadolinium oxide precursor, provides Gd³⁺ cations | Dried at 800 °C prior to use [7] |
| TiO₂ | Titanium oxide precursor; in MSS, acts as a template | Dried at 180 °C prior to use [7] |
| NaCl:KCl Eutectic | Molten salt medium (1:1 molar ratio); provides liquid phase for enhanced diffusion | Dried at 110 °C [7] |
| Isopropanol/Cyclohexane | Carrier fluid for milling/pre-mixing of precursors | - |
- Precursor Preparation: Dry Gd₂O₃ at 800 °C and TiO₂ at 180 °C overnight. Weigh stoichiometric amounts to yield a 10 g batch of Gd₂TiO₅.
- Salt and Mixture Preparation: Dry an equimolar NaCl:KCl eutectic mixture at 110 °C. Combine the salts with the precursor oxides in a 7:1 molar ratio of salt to ceramic precursor.
- Milling: Mill the mixture in a planetary ball mill at a low rotational frequency (250 rpm) for a short duration (5 minutes) using cyclohexane as a carrier fluid. This step mixes the components without significant particle size reduction.
- Pelletizing: Uniaxially press the powder mixture into pellets (e.g., 13 mm diameter) under 3 tonnes of pressure for 1 minute.
- Reaction: Fire the green pellets at the target temperature (e.g., 1300 °C) for 2 hours in air, using a heating and cooling rate of 5 °C/min.
- Purification: After reaction, wash the product with ultrapure water to dissolve and remove the water-soluble salt flux. Collect the purified Gd₂TiO₅ powder via vacuum filtration and dry.
- Precursor Preparation: Dry Gd₂O₃ and TiO₂ as described for MSS. Weigh stoichiometric amounts for a 10 g batch.
- Milling: Mill the precursor mixture in a planetary ball mill at a higher rotational frequency (500 rpm) for a longer duration (15 minutes) using isopropanol as the carrier fluid. This intensive milling is required to improve homogeneity.
- Drying and Sieving: Dry the resulting slurry at 80 °C overnight and pass the powder through a 250 μm sieve.
- Pelletizing: Press the powder into pellets as described in the MSS protocol.
- Reaction: Fire the green pellets at high temperature (e.g., 1500 °C) for 24 hours in air, using a heating and cooling rate of 5 °C/min. Multiple cycles of milling and firing are often needed to achieve a homogeneous product.
The choice between molten salt synthesis and conventional solid-state synthesis for Gd₂TiO₅ presents a clear trade-off between morphological control and procedural simplicity. MSS is unequivocally superior for applications requiring specific particle morphology, a narrow size distribution, and high phase purity at lower processing temperatures. The template growth mechanism inherent to MSS enables the direct production of fine, faceted particles around 1 μm. In contrast, SSS produces larger, more irregular particles and often struggles with phase purity without prolonged high-temperature treatment. The decision for researchers should be guided by the application-driven requirements for the final ceramic powder, with MSS offering a powerful route for advanced morphology control.
Optimizing Thermal Profiles and Reagent Preparation
The synthesis of complex ceramic materials, such as the 'stuffed' pyrochlore Gd₂TiO₅, presents significant challenges in solid-state chemistry. This material is of considerable interest for advanced nuclear applications, including as a matrix for actinide disposition and as a neutron absorber in control rods [7] [1]. The conventional solid-state synthesis (SSS) route for producing such refractory compounds typically requires prolonged high-temperature processing, which often results in undesirable microstructural characteristics and potential non-stoichiometry due to volatile component loss [7].
Molten Salt Synthesis (MSS) has emerged as a powerful alternative technique that addresses many limitations of traditional SSS. By utilizing a molten salt medium to enhance reactant diffusion, MSS enables the formation of phase-pure products at lower temperatures and significantly reduced reaction times [7] [13]. This comparative guide objectively examines both synthesis methods for Gd₂TiO₅, focusing specifically on the optimization of thermal profiles and reagent preparation parameters to achieve optimal product characteristics.
Methodological Comparison: MSS vs. SSS
Fundamental Synthesis Mechanisms
The two methods operate on fundamentally different reaction mechanisms, which directly impact their efficiency and the characteristics of the final product.
Solid-State Synthesis (SSS): This conventional approach relies on direct solid-state diffusion between metal oxide precursors. The process requires repeated milling and high-temperature treatments over extended durations (typically 24 hours or more) to facilitate atomic diffusion across particle boundaries [7]. This method often results in larger particle sizes (typically 2-3 μm) with angular morphology and may retain trace impurity phases even after prolonged heating at 1500°C [7].
Molten Salt Synthesis (MSS): This method utilizes a low-melting-point salt eutectic (commonly NaCl:KCl) that creates a liquid reaction medium at elevated temperatures. The process typically follows a template growth mechanism, where one reactant (TiO₂) with low solubility acts as a template while the more soluble reactant (Gd₂O₃) dissolves in the molten salt and diffuses to the template surface to react [7]. This mechanism enables the formation of products with controlled morphology and smaller particle sizes (~1 μm) at significantly reduced temperatures and reaction times [7].
Table 1: Comparative Overview of Synthesis Mechanisms and Outcomes
| Parameter | Molten Salt Synthesis (MSS) | Conventional Solid-State Synthesis (SSS) |
|---|---|---|
| Reaction Medium | Molten salt (NaCl:KCl eutectic) | Solid-state direct contact |
| Primary Mechanism | Template growth/solution-diffusion [7] | Solid-state diffusion [7] |
| Dominant Process | One reactant dissolves and diffuses to second reactant surface [7] | Atomic diffusion across particle boundaries [7] |
| Particle Morphology | Faceted, similar to TiO₂ template [7] | Angular, irregular [7] |
| Typical Particle Size | ~1 μm [7] | 2-3 μm [7] |
Experimental Protocols
Molten Salt Synthesis Protocol
The MSS pathway for Gd₂TiO₅ follows a meticulously optimized procedure [7]:
Reagent Preparation:
- Pre-dry Gd₂O₃ at 800°C and TiO₂ at 180°C overnight to remove adsorbed moisture
- Batch stoichiometric amounts of reagents (typically 10g total)
Salt and Precursor Mixing:
- Utilize NaCl:KCl eutectic mixture (1:1 molar ratio) dried at 110°C
- Maintain 7:1 molar ratio of molten salt to ceramic precursor
- Employ low-energy milling (250 rpm for 5 minutes) with cyclohexane as carrier fluid to minimize particle size reduction
Reaction Configuration:
- Uniaxially press mixture into 13mm pellets (3 tonnes for 1 minute)
- Heat at 5°C/min to target temperature (700-1500°C)
- Maintain at peak temperature for 2 hours
- Cool at controlled rate (5°C/min)
Post-Synthesis Processing:
- Wash product with ultrapure water to remove salt medium
- Collect purified powder via vacuum filtration
Solid-State Synthesis Protocol
The conventional SSS approach follows a more energy-intensive pathway [7]:
Reagent Preparation:
- Employ identical pre-drying procedures for Gd₂O₃ and TiO₂
- Batch stoichiometric amounts (10g total)
Precursor Processing:
- Utilize high-energy planetary ball milling (500 rpm for 15 minutes)
- Use isopropanol as carrier fluid with Sialon pot and media
- Dry resulting slurry at 80°C overnight
- Pass dried powder through 250μm sieve
Reaction Configuration:
- Press 0.5g samples into 13mm pellets (3 tonnes for 1 minute)
- Heat at 5°C/min to target temperature (700-1500°C)
- Maintain at peak temperature for 24 hours
- Cool at controlled rate (5°C/min)
The following workflow diagram illustrates the comparative procedures for both synthesis methods:
Thermal Profile Optimization
Temperature and Duration Parameters
The thermal profile represents a critical differentiator between MSS and SSS approaches. Extensive comparative studies have quantified the significant advantages of MSS in reducing both reaction temperature and duration while achieving superior phase purity [7].
Table 2: Optimized Thermal Parameters for Gd₂TiO₅ Synthesis
| Synthesis Method | Optimal Temperature | Minimum Time | Heating/Cooling Rate | Phase Purity Outcome |
|---|---|---|---|---|
| Molten Salt Synthesis | 1300°C | 2 hours | 5°C/min | Single-phase Gd₂TiO₅ [7] |
| Solid-State Synthesis | 1500°C | 24 hours | 5°C/min | Trace Gd₂Ti₂O₇ and Gd₂O₃ remains [7] |
Phase Evolution Pathways
Both synthesis methods exhibit distinct phase evolution pathways during thermal processing:
MSS Pathway: At intermediate temperatures (800-900°C), the pyrochlore phase Gd₂Ti₂O₇ forms initially as a kinetic product alongside residual Gd₂O₃. As temperature increases to 1100°C, Gd₂TiO₅ becomes the predominant phase. By 1300°C, complete conversion to single-phase Gd₂TiO₅ occurs within just 2 hours [7].
SSS Pathway: Initial Gd₂TiO₅ formation begins at 900°C after 24-hour treatment, accompanied by Gd₂Ti₂O₇ and residual Gd₂O₃. While Gd₂TiO₅ becomes the major phase at 1000°C, trace amounts of Gd₂Ti₂O₇ and Gd₂O₃ persist even after 24-hour treatment at 1500°C, as evidenced by the (222) reflection of Gd₂O₃ at 2θ = 29° and (111) reflection of Gd₂Ti₂O₇ at 2θ = 15° [7].
The following diagram illustrates the comparative phase evolution for both synthesis methods:
Reagent Preparation and Formulation
Research Reagent Solutions
The preparation and selection of reagents significantly influence the synthesis outcome for both MSS and SSS approaches. The following table details the essential materials and their specific functions in the synthesis processes:
Table 3: Essential Research Reagents for Gd₂TiO₅ Synthesis
| Reagent | Function | Preparation Protocol | Critical Parameters |
|---|---|---|---|
| Gd₂O₃ | Oxide precursor providing Gd cations | Dry at 800°C overnight to remove adsorbed H₂O and CO₂ [7] | High purity (>99%), controlled moisture content |
| TiO₂ | Oxide precursor providing Ti cations; acts as template in MSS | Dry at 180°C overnight [7] | Particle morphology determines final product morphology in MSS |
| NaCl:KCl Eutectic | Molten salt medium for MSS (1:1 molar ratio) | Dry at 110°C [7] | 7:1 molar ratio of salt to ceramic precursor [7] |
| Isopropanol | Carrier fluid for high-energy milling in SSS | Use as-received analytical grade | Prevents overheating during milling |
| Cyclohexane | Carrier fluid for low-energy milling in MSS | Use as-received analytical grade | Minimizes particle size reduction during mixing |
Molten Salt Selection Criteria
The choice of molten salt medium represents a critical optimization parameter in MSS. The NaCl:KCl eutectic system offers several distinct advantages:
- Low Eutectic Temperature: The 1:1 molar ratio NaCl:KCl mixture melts at approximately 650°C, creating a liquid reaction medium well below the synthesis temperature [13]
- Optimal Ion Mobility: The molten state provides enhanced ion mobility compared to solid-state diffusion, accelerating reaction kinetics
- Water Solubility: The high water solubility of both NaCl and KCl enables facile removal through washing with ultrapure water [7]
- Chemical Inertness: These salts do not participate in the reaction, serving solely as a liquid medium [7]
milling Energy Optimization
The milling parameters differ significantly between MSS and SSS, reflecting their distinct mechanistic requirements:
- SSS Requirements: High-energy planetary ball milling (500 rpm for 15 minutes) necessary to maximize interfacial contact between reactant particles for solid-state diffusion [7]
- MSS Requirements: Low-energy milling (250 rpm for 5 minutes) sufficient for homogenization while preserving template morphology for the template growth mechanism [7]
Performance Comparison and Applications
Synthesis Efficiency Metrics
The quantitative advantages of MSS over SSS become evident when comparing key efficiency metrics:
- Energy Consumption: MSS reduces thermal energy input by approximately 50% through lower temperature (1300°C vs. 1500°C) and shorter duration (2h vs. 24h) [7]
- Temporal Efficiency: MSS achieves complete reaction 12 times faster than SSS (2h vs. 24h) at optimal temperatures [7]
- Phase Purity: MSS produces single-phase Gd₂TiO₅, while SSS retains trace impurities even after extended heating [7]
- Particle Size Control: MSS enables finer particle sizes (~1μm) compared to SSS (2-3μm) [7]
Application-Specific Recommendations
Based on the comparative performance data, specific recommendations emerge for different research applications:
- Nuclear Materials Research: MSS is strongly recommended for Gd₂TiO₅ synthesis due to its superior phase purity, which is critical for accurate radiation damage studies and nuclear waste form development [7] [1]
- Structure-Property Studies: MSS provides more controlled morphology and reduced particle size, enabling more precise correlation between structure and properties
- Scale-Up Considerations: MSS offers potential economic advantages for industrial-scale production due to reduced energy requirements and processing times
- Fundamental Kinetics Studies: SSS may be preferred for investigating solid-state reaction mechanisms without the complicating factor of a liquid medium
The optimization of thermal profiles and reagent preparation strategies reveals clear advantages for Molten Salt Synthesis in the preparation of Gd₂TiO₅ 'stuffed' pyrochlore. MSS enables the formation of single-phase material at significantly reduced temperatures (1300°C vs. 1500°C) and dramatically shorter reaction times (2 hours vs. 24 hours) through a template growth mechanism facilitated by the molten salt medium [7].
The critical optimization parameters for MSS include the use of NaCl:KCl eutectic mixture with 7:1 salt-to-precursor ratio, controlled low-energy milling to preserve template morphology, and precisely regulated thermal profiles with 5°C/min heating and cooling rates. These parameters collectively enable the production of phase-pure Gd₂TiO₅ with controlled particle morphology and size distribution, representing a significant advancement over conventional solid-state approaches for this technologically important refractory material.
For researchers pursuing nuclear applications of Gd₂TiO₅, where phase purity and microstructural control are paramount, MSS represents the clearly superior synthesis methodology based on the comprehensive experimental data presented in this comparison.
Head-to-Head Comparison: Efficiency, Purity, and Microstructure
The pursuit of single-phase, high-purity ceramic materials is fundamental to advanced research and development across numerous scientific disciplines. For refractory ceramics such as gadolinium titanate (Gd₂TiO₅)—a material of significant interest for nuclear applications and functional materials—the synthesis pathway critically determines the phase purity and crystalline quality of the final product [7]. This guide provides an objective comparative analysis of two principal synthesis methods: Molten Salt Synthesis (MSS) and conventional Solid-State Synthesis (SSS). By presenting quantitative X-ray diffraction (XRD) data and detailed experimental protocols, we aim to equip researchers with the information necessary to select the most appropriate synthesis method for their specific application requirements.
Synthesis Methodologies and XRD Comparative Analysis
Experimental Protocols
Molten Salt Synthesis (MSS) Protocol [7]
- Precursor Preparation: Gd₂O₃ and TiO₂ reagents are dried overnight at 800°C and 180°C, respectively. Stoichiometric amounts are batched for a 10g final product.
- Flux Addition: An equimolar eutectic mixture of NaCl:KCl is added at a 7:1 molar ratio of molten salt to ceramic precursor.
- Mixing: The salt and precursor mixture is planetary ball-milled at 250 rpm for 5 minutes using cyclohexane as a carrier fluid to minimize size reduction.
- Pelletization: The mixed powder is uniaxially pressed into 13 mm pellets under 3 tonnes of pressure.
- Reaction: Pellets are reacted at temperatures between 700–1500°C (in 100°C intervals) for a short duration of 2 hours, with heating and cooling rates of 5°C/min.
- Purification: The cooled product is washed with ultrapure water to remove the salt flux, and the final powder is collected via vacuum filtration.
Solid-State Synthesis (SSS) Protocol [7]
- Precursor Preparation: Gd₂O₃ and TiO₂ are dried as in the MSS protocol. Stoichiometric amounts are batched for a 10g final product.
- Milling: The precursor mixture is planetary ball-milled at 500 rpm for 15 minutes using isopropanol as a carrier fluid.
- Drying and Sieving: The resulting slurry is dried at 80°C overnight and passed through a 250 μm sieve.
- Pelletization: 0.5 g samples are pressed into 13 mm pellets under 3 tonnes of pressure.
- Reaction: Green pellets are reacted at temperatures between 700–1500°C (in 100°C intervals) for a long duration of 24 hours, with heating and cooling rates of 5°C/min.
Workflow Diagram
The following diagram illustrates the key procedural steps and fundamental mechanisms that differentiate the two synthesis methods.
Quantitative XRD Data Comparison
XRD analysis provides a direct comparison of the phase purity and synthesis efficiency of Gd₂TiO₅ produced by both methods. The table below summarizes the quantitative findings.
Table 1: Comparative XRD Analysis of Gd₂TiO₅ Synthesis Methods [7]
| Synthesis Parameter | Molten Salt Synthesis (MSS) | Solid-State Synthesis (SSS) |
|---|---|---|
| Optimal Temperature & Time | 1300°C for 2 hours | 1500°C for 24 hours |
| Phase Purity at Optimal Condition | Single-phase Gd₂TiO₅ (No secondary phases detected) | Major phase Gd₂TiO₅ with trace Gd₂Ti₂O₇ and Gd₂O₃ |
| Initial Phase Formation | 800–900°C | 900°C |
| Primary Reaction Mechanism | Template growth (Solution-Diffusion) | Solid-State Diffusion |
| Particle Size (from SEM) | ~1 μm (similar to TiO₂ precursor) | 2–3 μm (larger, angular particles) |
| Particle Morphology | Faceted, uniform | Angular, sintered |
The Scientist's Toolkit: Essential Research Reagents and Materials
The successful execution of these synthesis protocols relies on specific reagents and instrumentation. The following table details the key materials and their functions.
Table 2: Essential Research Reagents and Instrumentation for Gd₂TiO₅ Synthesis and Characterization [7]
| Item/Category | Specific Function in Synthesis & Characterization |
|---|---|
| Gd₂O₃ (Gadolinium Oxide) | Gadolinium precursor. Must be pre-dried at 800°C to remove adsorbed water. |
| TiO₂ (Titanium Dioxide) | Titanium precursor. Must be pre-dried at 180°C. Acts as a template in MSS. |
| NaCl:KCl Eutectic Flux | Molten salt medium in MSS. Lowers synthesis temperature by facilitating reactant diffusion. |
| Planetary Ball Mill | For homogenizing and intimately mixing reactant powders before reaction. |
| High-Temperature Furnace | For achieving reaction temperatures up to 1500°C under air atmosphere. |
| X-ray Diffractometer (XRD) | Primary tool for phase identification, quantification, and purity assessment. |
| Rietveld Refinement Software | Standardless method for quantitative phase analysis (QPA) from XRD patterns [16]. |
The comparative XRD data unequivocally demonstrates that Molten Salt Synthesis offers significant advantages over Solid-State Synthesis for the production of Gd₂TiO₅. The MSS protocol achieves superior phase purity at a lower temperature (1300°C) and with a drastically reduced reaction time (2 hours vs. 24 hours). The "template growth" mechanism in MSS not only enhances kinetics but also results in smaller, more uniform particles. For research applications where high phase purity, energy efficiency, and fine particle size are critical, MSS is the recommended method. Conversely, Solid-State Synthesis, while conceptually simpler, requires higher temperatures and prolonged processing, yet may still yield products with detectable impurity phases, making it less suitable for applications demanding absolute phase purity.
In the field of inorganic materials synthesis, the pursuit of efficient reaction pathways is paramount. This guide provides a comparative analysis of synthesis methods for Gd₂TiO₅ 'stuffed' pyrochlore, a refractory material of significant interest for nuclear applications, including actinide disposition matrices and neutron absorbers in control rods [7]. The reaction kinetics—specifically the interplay between temperature and time—serves as a critical determinant of synthesis efficiency, product purity, and practical feasibility. Within the broader thesis of molten salt solid-state synthesis research, this article objectively evaluates the performance of Molten Salt Synthesis (MSS) against Conventional Solid-State Synthesis (SSS) using quantitative experimental data, detailing methodologies and providing visualization of the underlying processes.
Comparative Experimental Data: MSS vs. SSS
The synthesis of single-phase Gd₂TiO₅ proceeds through an intermediate kinetic product, Gd₂Ti₂O₇ pyrochlore [7]. The quantitative outcomes for each method, under optimized conditions, are summarized in Table 1.
Table 1: Quantitative Comparison of Gd₂TiO₅ Synthesis Methods
| Synthesis Parameter | Molten Salt Synthesis (MSS) | Conventional Solid-State Synthesis (SSS) |
|---|---|---|
| Optimal Temperature | 1300 °C | >1500 °C |
| Optimal Time | 2 hours | 24 hours |
| Final Product Purity | Single-phase Gd₂TiO₅ | Gd₂TiO₅ with trace Gd₂Ti₂O₇ and Gd₂O₃ |
| Intermediate Phase | Gd₂Ti₂O₇ (formed at 800-900 °C) | Gd₂Ti₂O₇ (formed at 900 °C) |
| Particle Morphology | Faceted, ~1 μm | Angular, 2-3 μm |
| Proposed Mechanism | Template growth | Solid-state diffusion |
The data reveals that MSS achieves a kinetically favorable pathway, reducing both the required temperature and time while yielding a superior phase-pure product [7] [1]. The formation of the intermediate Gd₂Ti₂O₇ phase is a common feature in both processes, but its subsequent conversion to the final 'stuffed' Gd₂TiO₅ structure is markedly more efficient in the molten salt medium.
Detailed Experimental Protocols
Molten Salt Synthesis (MSS) of Gd₂TiO₅
1. Precursor Preparation:
- Reagents: Gd₂O₃ and TiO₂ (anatase or rutile).
- Pre-treatment: Dry Gd₂O₃ at 800 °C and TiO₂ at 180 °C overnight to remove adsorbed water.
- Stoichiometry: Batch stoichiometric amounts to yield a final composition of Gd₂TiO₅.
2. Flux Addition and Milling:
- Salt Flux: Use an equimolar eutectic mixture of NaCl and KCl. Dry the salt mixture at 110 °C prior to use.
- Salt-to-Precursor Ratio: Employ a 7:1 molar ratio of molten salt to ceramic precursor [7].
- Milling: Combine precursors and salt flux, then mill using a planetary ball mill at 250 rpm for 5 minutes. Use cyclohexane as a carrier fluid to minimize size reduction of the starting reagents.
3. Pelletization and Reaction:
- Forming: Uniaxially press the mixed powder into pellets (e.g., 13 mm diameter) under 3 tonnes of pressure for 1 minute.
- Thermal Profile: Heat the pellets to a target temperature of 1300 °C at a rate of 5 °C/min. Hold at this temperature for 2 hours, then cool at 5 °C/min.
4. Post-Synthesis Processing:
- Washing: After reaction, wash the product repeatedly with ultrapure water to dissolve and remove the NaCl/KCl flux.
- Recovery: Recover the purified Gd₂TiO₅ powder via vacuum filtration and drying [7].
Conventional Solid-State Synthesis (SSS) of Gd₂TiO₅
1. Precursor Preparation:
- Reagents: The same pre-treated Gd₂O₃ and TiO₂ used in MSS.
- Milling: Mill the precursor mixture in a planetary ball mill at 500 rpm for 15 minutes, using isopropanol as a carrier fluid.
- Drying and Sieving: Dry the resulting slurry at 80 °C overnight and pass the powder through a 250 μm sieve.
2. Pelletization and Reaction:
- Forming: Press the powder into pellets as described in the MSS protocol.
- Thermal Profile: Heat the pellets to temperatures exceeding 1500 °C (e.g., in 100 °C intervals from 700 °C to 1500 °C) with a heating rate of 5 °C/min. A typical dwell time at the maximum temperature is 24 hours to approach completion, though trace impurities may persist [7].
Visualization of Synthesis Pathways and Mechanisms
The distinct reaction kinetics and mechanisms of the two synthesis methods are illustrated below.
The diagram highlights the more complex, liquid-mediated pathway of MSS, which facilitates faster kinetics at lower temperatures. The "template growth" mechanism is a key differentiator. In this process, the TiO₂ particles, which have low solubility in the alkali chloride salt, act as a physical template. The Gd₂O₃, which is more soluble, dissolves into the molten salt and diffuses to the surface of the TiO₂ particles where it reacts to form the product, ultimately preserving the morphology of the TiO₂ template [7]. This provides a direct and rapid reaction pathway.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents and Materials for Gd₂TiO₅ Synthesis
| Reagent/Material | Function in the Synthesis | Notes for Researchers |
|---|---|---|
| Gadolinium Oxide (Gd₂O₃) | Lanthanide cation precursor for the Gd₂TiO₅ crystal structure. | Must be pre-dried at 800 °C to ensure stoichiometric accuracy [7]. |
| Titanium Dioxide (TiO₂) | Source of Ti cations for the pyrochlore structure. | Pre-dry at 180 °C. Particle morphology may influence final product in MSS [7]. |
| Sodium Chloride (NaCl) | Component of the low-melting eutectic flux in MSS. | Use high-purity, anhydrous grade. Forms a eutectic with KCl (mp ~657 °C) [7]. |
| Potassium Chloride (KCl) | Component of the low-melting eutectic flux in MSS. | Use high-purity, anhydrous grade. Critical for creating the liquid reaction medium [7]. |
| Isopropanol / Cyclohexane | Carrier fluid for milling and mixing of precursors. | Prevents agglomeration and ensures homogeneity. Cyclohexane is preferred in MSS to minimize reagent dissolution [7]. |
Theoretical Context: Thermodynamic vs. Kinetic Control
The efficiency of MSS can be understood through the lens of solid-state reaction control. Recent research has quantified a threshold for thermodynamic control, where the initial product formed is the one with the largest thermodynamic driving force (max-ΔG), provided its driving force exceeds that of all competing phases by ≥60 milli–electron volt per atom (meV/atom) [17].
In SSS, with its high activation barriers and slow solid-state diffusion, reactions often fall under kinetic control, where the first phase to form may be a metastable intermediate (like Gd₂Ti₂O₇) due to lower nucleation barriers, even if it is not the most thermodynamically stable product. The MSS method, by creating a liquid medium that enhances ion mobility, can push the reaction into a regime closer to thermodynamic control. The liquid salt reduces kinetic limitations, allowing the system to bypass prolonged intermediate stages and form the stable Gd₂TiO₅ phase more directly and rapidly [7] [17]. This fundamental difference explains the dramatic improvements in temperature and time efficiency observed in the experimental data.
The synthesis of advanced ceramic materials, such as gadolinium titanate (Gd₂TiO₅), is pivotal for applications ranging from nuclear reactor control rods to phosphors in solid-state lighting [7] [11] [18]. The microstructure of these materials—including particle size, morphology, and phase distribution—is directly influenced by the synthesis method and dictates the final product's performance properties. This guide objectively compares the microstructural evolution of Gd₂TiO₅ fabricated via two distinct synthetic pathways: Molten Salt Synthesis (MSS) and Conventional Solid-State Synthesis (SSS), providing researchers with a data-driven framework for selecting an appropriate synthesis technique.
Experimental Protocols and Methodologies
Molten Salt Synthesis (MSS) of Gd₂TiO₅
The MSS process utilizes a molten salt medium to enhance diffusion and accelerate reaction kinetics at lower temperatures compared to solid-state routes [7].
- Precursor Preparation: Gd₂O₃ and TiO₂ reagents are dried overnight at 800 °C and 180 °C, respectively. Stoichiometric amounts are batched to achieve the target Gd₂TiO₅ composition [7].
- Salt Flux Addition: An equimolar eutectic mixture of NaCl and KCl (1:1 mol ratio) is used as the flux. A salt-to-ceramic precursor ratio of 7:1 mol is employed [7].
- Mixing: The salt and precursor mixture is milled together in a planetary ball mill at 250 rpm for 5 minutes using cyclohexane as a carrier fluid to minimize particle size reduction [7].
- Reaction: The milled mixture is uniaxially pressed into pellets and reacted at temperatures between 700 °C and 1500 °C for 2 hours, with heating and cooling rates of 5 °C/min [7].
- Purification: The reacted product is washed with ultrapure water to remove the soluble salt flux, and the resulting powder is collected via vacuum filtration [7].
Conventional Solid-State Synthesis (SSS) of Gd₂TiO₅
The SSS method relies on direct solid-state diffusion between oxide precursors, typically requiring higher temperatures and longer durations [7] [9].
- Precursor Preparation: Gd₂O₃ and TiO₂ are similarly dried. Stoichiometric amounts are batched for a 10 g total output [7].
- Milling: The precursor mixture is planetary ball-milled at 500 rpm for 15 minutes using isopropanol as a carrier fluid [7].
- Pelletization: The resulting powder slurry is dried, sieved through a 250 μm mesh, and uniaxially pressed into pellets [7].
- Reaction: Pellets are reacted at temperatures from 700 °C to 1500 °C for 24 hours at each temperature interval, with heating and cooling rates of 5 °C/min [7]. Other studies report even longer durations, such as 24 hours at 1300 °C, to achieve a pure phase [9].
Characterization Techniques
- X-ray Diffraction (XRD): Used for phase identification and confirmation of crystal structure (orthorhombic for Gd₂TiO₅, space group Pnma) [7] [11].
- Scanning Electron Microscopy (SEM): Employed to analyze particle size, morphology, and surface features. Samples are typically dispersed in acetone, ultrasonicated to break agglomerates, and deposited on a carbon stub prior to imaging [7].
Comparative Microstructural and Morphological Analysis
The synthesis method profoundly impacts the phase purity, particle size, and morphology of the final Gd₂TiO₅ product, as summarized in the table below.
Table 1: Quantitative Comparison of Gd₂TiO₅ Synthesized via MSS and SSS
| Analysis Parameter | Molten Salt Synthesis (MSS) | Conventional Solid-State Synthesis (SSS) |
|---|---|---|
| Optimal Synthesis Conditions | 1300 °C for 2 hours [7] | >1300 °C for 24 hours or more [7] [9] |
| Phase Purity at Optimal Conditions | Single-phase Gd₂TiO5 achieved [7] | Trace amounts of Gd₂Ti₂O7 and Gd₂O3 often persist even at 1500 °C [7] |
| Average Particle Size | ~1 μm [7] | 2–3 μm [7] |
| Particle Morphology | Faceted, well-defined particles [7] | Larger, more angular, and irregular particles [7] |
| Primary Formation Mechanism | Template growth (Solution-Diffusion) [7] | Solid-state diffusion and sintering [7] |
Interpretation of Morphological Differences
The stark differences in microstructure arise from the fundamental mechanisms governing each synthesis route.
- Mechanism in MSS: The MSS process follows a template growth mechanism [7]. In this process, TiO₂ particles, which have low solubility in the alkali chloride salt melt, act as a template. The Gd₂O₃, which is more soluble, dissolves into the molten salt, diffuses to the surface of the TiO₂ particles, and reacts to form the product. This mechanism explains the similarity in size and faceted morphology between the final Gd₂TiO₅ particles and the original TiO₂ reagents [7].
- Mechanism in SSS: The SSS process is governed by solid-state diffusion [7]. This process requires high temperatures and long durations for atomic diffusion across particle boundaries, leading to coarsening and grain growth. This results in larger, more sintered, and angular particles compared to those produced by MSS [7].
The following workflow diagrams illustrate the distinct synthetic pathways and their resulting microstructures.
Diagram 1: A comparative workflow of MSS and SSS pathways for Gd₂TiO₅.
The Scientist's Toolkit: Key Research Reagents
The following table details essential materials and their functions in the synthesis and characterization of Gd₂TiO₅.
Table 2: Essential Research Reagents for Gd₂TiO₅ Synthesis and Analysis
| Reagent / Material | Function in Research | Application Context |
|---|---|---|
| Gadolinium Oxide (Gd₂O₃) | Primary gadolinium source precursor | MSS & SSS [7] [9] |
| Titanium Dioxide (TiO₂) | Primary titanium source precursor; acts as a template in MSS | MSS & SSS [7] [9] |
| Sodium Chloride (NaCl) | Component of low-melting-point eutectic flux | MSS (as NaCl:KCl mixture) [7] |
| Potassium Chloride (KCl) | Component of low-melting-point eutectic flux | MSS (as NaCl:KCl mixture) [7] |
| Dysprosium/Europium Nitrates | Dopant sources for photoluminescence studies | Sol-gel synthesis of phosphors [11] |
| Citric Acid | Chelating agent in sol-gel synthesis | Sol-gel method [11] |
The choice between Molten Salt Synthesis and Solid-State Synthesis for fabricating Gd₂TiO₅ presents a clear trade-off. MSS offers significant advantages in microstructural control, enabling the production of smaller, morphologically defined particles with high phase purity at lower temperatures and significantly shorter reaction times. This makes it superior for applications where specific surface area and particle size are critical. In contrast, SSS is a more straightforward, albeit energy-intensive, method that can be adequate for applications where larger particle size and the potential for minor secondary phases are not detrimental. The decision should be guided by the target properties and performance requirements of the final material in its intended application, whether for nuclear energy or advanced photonics.
Comparative Performance in Target Application Metrics
The synthesis of complex inorganic ceramics like gadolinium titanate (Gd₂TiO₅) is crucial for advancing applications in nuclear engineering, phosphor materials, and waste immobilization technologies. The choice of synthesis pathway directly influences critical performance metrics including phase purity, morphological characteristics, processing efficiency, and functional properties. This guide provides a systematic comparison between the conventional solid-state method and the emerging molten salt synthesis approach for Gd₂TiO₅, presenting experimental data to objectively evaluate their performance against target application requirements.
Experimental Protocols for Gd₂TiO₅ Synthesis
Molten Salt Synthesis (MSS) Protocol
Primary Reagents: Gd₂O₃, TiO₂, NaCl, KCl [7]. Synthesis Procedure:
- Precursor Preparation: Dry Gd₂O₃ at 800°C and TiO₂ at 180°C overnight to remove adsorbed moisture [7].
- Salt and Reactant Mixing: Combine oxide precursors with an equimolar eutectic mixture of NaCl:KCl (1:1 mol ratio). Use a 7:1 molar ratio of molten salt to ceramic precursor mixture [7].
- Milling: Mill the salt and precursor mixture using a planetary mill at 250 rpm for 5 minutes with cyclohexane as a carrier fluid to minimize size reduction [7].
- Pelletization: Uniaxially press the mixture into 13 mm pellets under 3 tonnes of pressure for 1 minute [7].
- Thermal Reaction: Heat pellets at 5°C/min to 1300°C and hold for 2 hours [7].
- Purification: Wash the reacted product with ultrapure water to remove the salt medium, then collect the final powder via vacuum filtration [7].
Solid-State Synthesis (SSS) Protocol
Primary Reagents: Gd₂O₃, TiO₂ [7] [9]. Synthesis Procedure:
- Precursor Preparation: Dry Gd₂O₃ at 800°C and TiO₂ at 180°C overnight [7].
- Stoichiometric Calculation: Batch stoichiometric amounts of reagents for 10g of final product [7].
- Milling: Mill precursors in a planetary ball mill (Pulverisette 6, Fritsch) at 500 rpm for 15 minutes using isopropanol as carrier fluid [7].
- Pelletization: Uniaxially press dried and sieved powder into 13 mm pellets under 3 tonnes of pressure for 1 minute [7].
- Thermal Reaction: Heat pellets at 5°C/min to 1300-1500°C and hold for 24 hours [7]. For phosphor applications, a three-step heating protocol may be employed with final sintering at 1300°C for 24 hours [9].
Comparative Performance Metrics
Synthesis Efficiency and Phase Purity
Table 1: Synthesis Efficiency Comparison Between MSS and SSS for Gd₂TiO₅
| Performance Metric | Molten Salt Synthesis (MSS) | Conventional Solid-State Synthesis (SSS) |
|---|---|---|
| Reaction Temperature | 1300°C [7] | 1300-1500°C [7] |
| Reaction Time | 2 hours [7] | 24 hours or more [7] [9] |
| Phase Purity Achievement | Single phase at 1300°C/2h [7] | Trace impurities (Gd₂Ti₂O₇, Gd₂O₃) even at 1500°C/24h [7] |
| Formation Pathway | Proceeds through Gd₂Ti₂O₇ intermediate via template growth mechanism [7] | Direct reaction from oxide precursors [7] |
| Particle Size Range | 50-500 nm [8] | 2-3 μm [7] |
| Particle Morphology | Faceted morphology similar to TiO₂ precursor [7] | Angular morphology with larger, irregular particles [7] |
Material Characteristics and Application Performance
Table 2: Material Characteristics and Functional Performance of Gd₂TiO₅
| Characteristic | Synthesis Method | Performance Metrics | Application Implications |
|---|---|---|---|
| Phase Purity | MSS | Single phase achievable at 1300°C/2h [7] | Preferred for nuclear applications requiring precise stoichiometry |
| Photoluminescence Performance | SSS (Gd₂TiO₅:Eu³⁺) | No concentration quenching up to 10 mol% Eu³⁺; PLQY ~14%; color purity ~99% [9] | Optimal for phosphor applications in solid-state lighting |
| Particle Size Control | MSS | 50-500 nm range achievable [8] | Beneficial for catalytic applications and sintering |
| Dopant Incorporation | SSS | Complete europium solubility with minimal lattice strain [9] | Ideal for high-performance phosphor design |
| Processing Efficiency | MSS | 92% reduction in reaction time vs. SSS [7] | Significant energy savings for industrial production |
Synthesis Workflow and Reaction Pathways
The following diagram illustrates the comparative synthesis pathways for Gd₂TiO₅ production:
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for Gd₂TiO₅ Synthesis and Characterization
| Reagent/Material | Function/Purpose | Specifications |
|---|---|---|
| Gadolinium Oxide (Gd₂O₃) | Primary gadolinium source | 99.9% purity, pre-dried at 800°C [7] [9] |
| Titanium Dioxide (TiO₂) | Primary titanium source | 99.0-99.9% purity, pre-dried at 180°C [7] [9] |
| NaCl:KCl Eutectic | Molten salt medium for MSS | 1:1 molar ratio, melting point 650°C [7] |
| Europium Oxide (Eu₂O₃) | Photoluminescent dopant | 99.0% purity for phosphor applications [9] |
| Isopropanol/Cyclohexane | Milling carrier fluid | Prevents agglomeration during particle size reduction [7] |
Application-Specific Performance Recommendations
Nuclear Applications
For nuclear applications including control rod materials and waste forms, molten salt synthesis is superior due to its ability to produce phase-pure Gd₂TiO₅ with minimal processing time and energy input [7]. The method's efficiency in achieving single-phase material at lower thermal budgets addresses key manufacturing constraints while maintaining the required structural integrity for nuclear service environments.
Photoluminescence Applications
For phosphor applications such as Gd₂TiO₅:Eu³⁺ in solid-state lighting, conventional solid-state synthesis remains preferred despite longer processing times. The method demonstrates exceptional capability in achieving complete dopant solubility without concentration quenching, high color purity (~99%), and satisfactory photoluminescence quantum yield (~14%) [9]. The extended reaction times facilitate optimal crystallinity and dopant incorporation essential for luminescent performance.
Catalytic and Advanced Ceramic Applications
For applications requiring specific surface area or particle size control, molten salt synthesis provides significant advantages through its template growth mechanism, producing faceted particles in the 50-500 nm range [7] [8]. The method's ability to control morphology through precursor selection offers additional tuning parameters for surface-sensitive applications.
Conclusion
This comparative analysis unequivocally demonstrates that molten salt synthesis offers a superior pathway for fabricating Gd₂TiO₅ compared to conventional solid-state methods, achieving high phase purity at lower temperatures and significantly reduced reaction times through a template growth mechanism. The efficiency and control afforded by MSS present profound implications for biomedical and clinical research, particularly in developing advanced materials for nuclear medicine, radiation shielding, and targeted therapeutic delivery systems. Future research should focus on optimizing salt chemistries for specific biomedical applications, scaling MSS protocols for industrial production, and exploring the doping of Gd₂TiO₅ with therapeutic isotopes to create novel theranostic platforms, thereby bridging advanced materials synthesis with cutting-edge clinical applications.
References
- 1. A preliminary investigation of the molten salt mediated ... [https://eprints.whiterose.ac.uk/id/eprint/174552/]
- 2. Gd 2 TiO 5 Crystal Structure [https://materials.springer.com/isp/crystallographic/docs/sd_1626495]
- 3. Gd 2 TiO 5 A (Gd 2 TiO 5 rt) Crystal Structure [https://materials.springer.com/isp/crystallographic/docs/sd_1838836]
- 4. Crystal structures of Ln2TiO5 (Ln = Gd, Dy) polymorphs [https://www.semanticscholar.org/paper/Crystal-structures-of-Ln2TiO5-(Ln-%3D-Gd%2C-Dy)-Shepelev-Petrova/e47517e9175843e9e1457af53f7a1d5c11f6d5f5]
- 5. Firing salts method for the synthesis of orthorhombic... | CoLab [https://colab.ws/articles/10.1088%2F2053-1591%2Fad4e0b]
- 6. and first-principles investigation on the optical properties... Experimental [https://link.springer.com/article/10.1007/s10854-025-14543-2]
- 7. A preliminary investigation of the molten salt mediated ... [https://link.springer.com/article/10.1557/s43580-021-00057-6]
- 8. Firing salts method for the synthesis of orthorhombic ... [https://doaj.org/article/0a1cdadbdfe642f8ab8128a6a63fdbbb]
- 9. High concentration quenching, improved PLQY and ... [https://www.sciencedirect.com/science/article/abs/pii/S0925346723006316]
- 10. Structural and luminescent properties of a Cr 3+ /Sm 3+ ... [https://www.sciencedirect.com/org/science/article/pii/S2046206925001810]
- 11. Dy 3+ /Eu 3+ nanophosphors for indoor illuminations [https://www.sciencedirect.com/science/article/abs/pii/S0925838821014286]
- 12. Molten salt synthesis of titanate pyrochlore waste-forms [https://www.sciencedirect.com/science/article/abs/pii/S0272884215023457]
- 13. Spectroscopic characterization of iron bismuth (antimony/ ... [https://www.sciencedirect.com/science/article/abs/pii/S0272884221024561]
- 14. The effect of synthesis temperature on structural ... [https://cst.kipmi.or.id/journal/article/view/1279]
- 15. A graph-based network for predicting chemical reaction ... [https://www.nature.com/articles/s41467-021-23339-x]
- 16. Phase Quantification Using XRD [https://www.malvernpanalytical.com/en/products/measurement-type/phase-quantification]
- 17. Quantifying the regime of thermodynamic control for solid- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11221506/]
- 18. Synthesis, characterization and thermal expansion studies ... [https://www.sciencedirect.com/science/article/abs/pii/S004060310800172X]